nosocomial Infections:
Hospital-acquired infections are referred to as nosocomial infections. They are also referred to as "HAI's", or "hospital-acquired infections". Annually, there are about 1.75-3 million cases of nosocomial infections that occur of approximately 35 million patients admitted to the hospital. Treatment ranges from $4.5 billion to $15 billion, adding to the cost of health care. Some of these infections result in the death of hospitalized patients (mortality) or lead to complications (morbidity). Infection control programs and departments are part of the healthcare team, and their goal is to prevent the acquisition of infection by both patients and healthcare workers.
Most hospitals have surveillance programs to monitor the incidences of nosocomial infections, which is mandated by the Centers for Disease Control and Prevention (CDC). Their research has shown that the most common types of nosocomial infections include catheter-associated urinary tract infections (CAUTI) (most common at 33%), pneumonia (15%), surgical site infections (SSI) (15%), and bloodstream infections (sepsis) (13%), which are often due to central line-associated bloodstream infections (CLABSI). Another type of HAI is due to multidrug-resistant organisms (MDRO).
Nosocomial infections have a high mortality rate of 40-60%. The surgical and medical wards tend to have the highest rates of infection, whereas the pediatric and nursery wards tend to have the lowest. Most nosocomial infections are a result of the patient's own microflora (opportunistic). Factors for acquiring a nosocomial infection include patient susceptibility and underlying health, microbial virulence factors, and the nature of exposure to the infectious microbe.
Most hospitals have surveillance programs to monitor the incidences of nosocomial infections, which is mandated by the Centers for Disease Control and Prevention (CDC). Their research has shown that the most common types of nosocomial infections include catheter-associated urinary tract infections (CAUTI) (most common at 33%), pneumonia (15%), surgical site infections (SSI) (15%), and bloodstream infections (sepsis) (13%), which are often due to central line-associated bloodstream infections (CLABSI). Another type of HAI is due to multidrug-resistant organisms (MDRO).
Nosocomial infections have a high mortality rate of 40-60%. The surgical and medical wards tend to have the highest rates of infection, whereas the pediatric and nursery wards tend to have the lowest. Most nosocomial infections are a result of the patient's own microflora (opportunistic). Factors for acquiring a nosocomial infection include patient susceptibility and underlying health, microbial virulence factors, and the nature of exposure to the infectious microbe.
Urinary tract infections:
Urinary tract infections, or UTI's, are the most common type of nosocomial infection. The major causative agent is Gram-negative rods (bacilli), mostly the Enterobacteriaceae (now called Enterobacterales). Escherichia coli is the #1 causative agent of these infections. Other microbes that often cause UTI's include Gram-positive organisms, Candida species, and other fungi. The elderly, females, and immunocompromised patients are at highest risk for infection. Indwelling urinary catheters are the #1 cause of nosocomial UTI's.
Lung infections:
The most common nosocomial infections resulting in pneumonia include Gram-negative rods, Staphylococcus aureus, Moraxella catarrhalis, Streptococcus pneumoniae, and Haemophilus influenzae. Risk factors include being elderly, having an underlying chronic lung disease, aspirating a large volume of fluid or exudate, post-chest surgery, hospitalization in the ICU, and intubation.
surgical site infections:
About 4% of all surgical patients end up developing a surgical site infection. Of these, 50% develop after the individual has already left the hospital. The major bacterial causes include Gram-positive organisms such as Staphylococcus aureus, CNS, Enterococcus faecalis, Gram-negative rods, and Candida species. Risk factors include being elderly, obesity, infection that seeds to other areas through the bloodstream (bacteremia or sepsis), malnutrition, diabetes, extended stay in the hospital after a surgical procedure, extended surgical time or time between surgical prep and actual surgery, failure to treat the patient with prophylactic antibiotic therapy.
bloodstream infections:
Bloodstream infections are most often caused by coagulase-negative staphylococci (CNS), enterococci, S. aureus, MRSA, or Candida species. Risk factors include newborns, the elderly, having an underlying disease, being immunocompromised, being malnourished, loss of skin integrity, having an indwelling catheter, going home with an indwelling catheter, being in the ICU, or prolonged stay in the hospital.
infection control programs:
Infection control programs have a goal to detect and monitor hospital-acquired infections, as well as to prevent and control their spread and prevent and control epidemics or outbreaks. A team of multidisciplinary healthcare workers is designed to review charts on a daily basis and look for patterns in order to detect an outbreak as soon as possible. They educate healthcare providers in sterile and aseptic techniques and isolation precautions. They also identify the cases involved in an outbreak and investigate the cause.
This team of individuals falls under the Quality Department, and typically includes a Quality-leader MD, a Director of Quality, Quality Analysts, a Clinical Educator, and an Infection Preventionist, a Public or Employee Health Nurse, EVS, as well as the support of nurse leaders and directors.
This team of individuals falls under the Quality Department, and typically includes a Quality-leader MD, a Director of Quality, Quality Analysts, a Clinical Educator, and an Infection Preventionist, a Public or Employee Health Nurse, EVS, as well as the support of nurse leaders and directors.
modes of contact/transmission:
- Direct contact (contaminated fluids like dialysate, contaminated food, contaminated IV solutions)
- Indirect contact (patient-to-patient, healthcare worker-to-patient, a fomite like a towel, linen, phone, bedrail, chair, curtain, ceiling tile, mirror, faucet, nursing equipment, etc...)
- Droplet contact (inhalation of droplet, skin or mucous membrane contact with droplet)
- Airborne contact (inhalation of aerosolized particles)
- Vector-borne contact (vectors like mosquitoes carry the microbe, bedbugs, ticks, etc...)
precautions:
The CDC published guidelines in the 1970's to specify isolation precautions in hospitals. They include the following:
- Healthcare workers must wash their hands between caring for different patients and change their gloves between caring for different patients
- If private rooms are not available, patients should be segregated in semiprivate rooms
- Masks, gowns, and gloves need to be worn when caring for infected patients
- Contaminated articles like bed linens need to be bagged when removed from the room
- All isolation rooms must be cleaned when the patient is discharged
- Cards need to be placed on the patient's door that specifically detail the type of isolation precautions and instructions for visitors and healthcare workers
categories of isolation:
- Strict Isolation (for highly contagious diseases like the chicken pox, shingles, pneumonia, Lassa fever, etc...)
- Respiratory Isolation (measles, H. influenzae, N. meningitidis)
- Enteric Precautions (gastroenteritis, amoebic dysentery, Salmonella, Shigella, C. difficile, Norovirus, E. coli O157:H7, E.coli enteroaggregative, E. coli enteroinvasive)
- Contact Isolation (MRSA, History of MRSA, MDRO's)
- AFB Isolation (tuberculosis)
- Draining and Secretion Precautions (burns, conjunctivitis)
- Blood and Body Fluid Precautions (HIV/AIDS)
CDC Standard precautions of 1996:
In 1996, the CDC developed a system of Standard Precautions called "universal precautions". These apply to ALL patients and apply to blood, all body fluids, secretions, broken skin, and mucous membranes. It means to treat all blood and body fluids as if they are contaminated.
surveillance programs and cultures:
Most hospitals perform a routine screening of the nares, axilla, or groin area to determine if a patient is carrying MRSA. This is the most common surveillance culture. All steam and dry-heat autoclave sterilizers are checked once a week with a liquid spore suspension to make sure there is no contamination and that the equipment is properly functioning. Dialysis and other fluids are checked once a month with a Millipore Total Count Water Tester to make sure that the dialysate being used for dialysis is not contaminated.
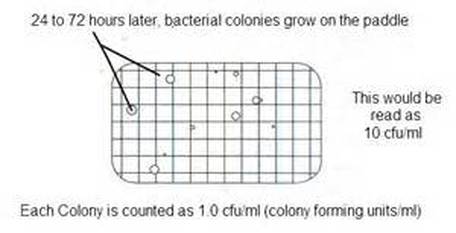
Steps How to Culture Hemodialysis Water and Dialysis Fluid:
Steps How to Culture Hemodialysis Water and Dialysis Fluid:
- The sampler is a self-contained device for sampling bacteria in water or fluid, contains an overlay with nutrient agar and it is sterile. Look to make sure that it is free from moisture and contamination prior to use.
- Before sampling, remove the paddle from the case and place it on a sterile piece of gauze under the biological safety cabinet.
- If you are testing dialysis fluid, it should be diluted 1:10 by filling the case with dialysate to the lower line, and filling the case to the upper line with sterile, distilled water.
- Add liquid to the case up to the top line marked on the case.
- Replace the paddle and secure it tightly.
- Shake the device several times.
- Set it down for 30 seconds to allow 1 mL of fluid to be absorbed onto the paddle. Once this is complete, the bubbling will stop from the little hole at the top of the case.
- Remove the paddle from the case and empty the fluid.
- Replace the paddle into the case and incubate the device, grid side up, at 35 degrees Celsius for 18-24 hours.
- Read the culture at 18-24 hours. If you are reading hemodialysis water, total viable counts should not exceed 200 colonies/mL. If you are reading dialysis fluid, total viable counts should not exceed 2,000 colonies/mL.
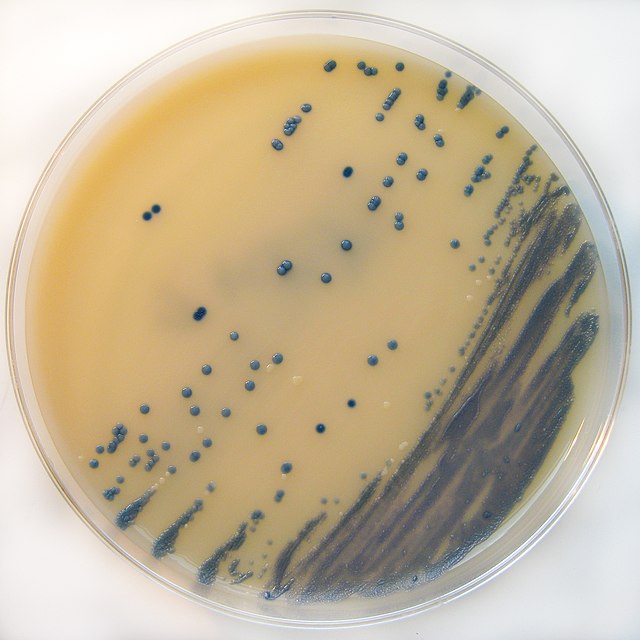
Salmonella Infection/Outbreak:
Salmonella spp are Gram-negative, motile rods. S. enterica is one of the most common strains that causes salmonellosis in humans.
Salmonella spp are never a part of the normal GI flora, so if they are isolated in the Microbiology laboratory, they are a true infection and may possibly be part of an outbreak. Salmonella causes diarrheal illness with stomach cramps and fever. Some strains cause Typhoid fever, but this site focuses on the strains that cause common diarrheal illness. The diarrhea may or may not be bloody in nature. Some people also experience nausea, vomiting and headache as well. Symptoms begin as early as 6 hours and up to 6 days after exposure, and they last 4-7 days. Diarrhea can lead to dehydration, bloodstream infection (bacteremia/sepsis), hospitalization, and UTI. Therefore, it may also be isolated in blood cultures, stool cultures, and urine cultures. It has also been associated with triggering post-infection reactive arthritis in some individuals. Infections are referred to as salmonellosis.
Laboratory testing involves sending stool samples to the lab in a clean container or in Cary-Blair preservative. Some laboratories, including the one where I work, test the stool on a GI panel PCR on the Biofire micro filmarray, which takes about an hour. Blood and urine samples are tested on the MALDI-TOF for identification, which also takes about an hour. In most states, including mine, this is a reportable organism to the state Public Health Laboratory. There, it is serotyped for the particular strain causing the infection, and then reported to other agencies which track cases and outbreaks.
Salmonella food poisoning is often caused by touching or ingesting contaminated eggs, poultry, beef, lettuce and vegetation, fruit, or eggs. It can also be caused by domestic pets, including cats, dogs, turtles, hamsters, guinea pigs, and other reptiles. Infections are more common during the summer months when it is warmer, there are more BBQ's and picnics, and people are consuming more summer fruits and veggies and out gardening. It is so important to wash one's hands after handling pets and dirt, and before eating.
Salmonella spp are never a part of the normal GI flora, so if they are isolated in the Microbiology laboratory, they are a true infection and may possibly be part of an outbreak. Salmonella causes diarrheal illness with stomach cramps and fever. Some strains cause Typhoid fever, but this site focuses on the strains that cause common diarrheal illness. The diarrhea may or may not be bloody in nature. Some people also experience nausea, vomiting and headache as well. Symptoms begin as early as 6 hours and up to 6 days after exposure, and they last 4-7 days. Diarrhea can lead to dehydration, bloodstream infection (bacteremia/sepsis), hospitalization, and UTI. Therefore, it may also be isolated in blood cultures, stool cultures, and urine cultures. It has also been associated with triggering post-infection reactive arthritis in some individuals. Infections are referred to as salmonellosis.
Laboratory testing involves sending stool samples to the lab in a clean container or in Cary-Blair preservative. Some laboratories, including the one where I work, test the stool on a GI panel PCR on the Biofire micro filmarray, which takes about an hour. Blood and urine samples are tested on the MALDI-TOF for identification, which also takes about an hour. In most states, including mine, this is a reportable organism to the state Public Health Laboratory. There, it is serotyped for the particular strain causing the infection, and then reported to other agencies which track cases and outbreaks.
Salmonella food poisoning is often caused by touching or ingesting contaminated eggs, poultry, beef, lettuce and vegetation, fruit, or eggs. It can also be caused by domestic pets, including cats, dogs, turtles, hamsters, guinea pigs, and other reptiles. Infections are more common during the summer months when it is warmer, there are more BBQ's and picnics, and people are consuming more summer fruits and veggies and out gardening. It is so important to wash one's hands after handling pets and dirt, and before eating.
campylobacter infection/outbreak:
Campylobacter infections are very similar to Salmonella infections and have the same cause, but the main natural reservoir for it is poultry. Infections result in gastroenteritis 24-72 hours after exposure. The symptoms are the same as with Salmonella, resulting in diarrhea that may or may not be bloody, cramps and pain, along with fever. Transmission is also via the fecal-oral route, by ingestion of contaminated food or water, usually by ingested contaminated undercooked meat. Raw or undercooked poultry, dairy products, and produce tend to be the sources of infection.
Campylobacter is a Gram-negative, anaerobic curved rod that is motile. It thrives at 42 degrees, and cells will die at 48 degrees or at freezing temperatures. It survives a short time at room temperature, which is why it should be in Cary-Blair preservative and refrigerated. It will survive 20x longer at 2 degrees than at 20 degrees, according to the CDC. Like Salmonella spp, it is a reportable organism to the state Public Health Laboratory, where it is sent, serotyped, recorded, and then sent to other agencies that track cases and outbreaks. C. jejuni and C. coli are the most common strains that cause infections in humans.
Campylobacter is a Gram-negative, anaerobic curved rod that is motile. It thrives at 42 degrees, and cells will die at 48 degrees or at freezing temperatures. It survives a short time at room temperature, which is why it should be in Cary-Blair preservative and refrigerated. It will survive 20x longer at 2 degrees than at 20 degrees, according to the CDC. Like Salmonella spp, it is a reportable organism to the state Public Health Laboratory, where it is sent, serotyped, recorded, and then sent to other agencies that track cases and outbreaks. C. jejuni and C. coli are the most common strains that cause infections in humans.
E. coli 0157:h7 infection/outbreak:
E. coli 0157:H7 is a toxogenic strain of E. coli that produces a shiga-like toxin that causes foodborne illness and severe gastroenteritis after consuming contaminated raw produce, dairy products, or by ingesting raw or undercooked contaminated beef. It can lead to bloody diarrhea and cramping, little to no fever, and even kidney failure and secondary hemolytic uremic syndrome (HUS) in susceptible individuals. It can be deadly. Transmission is via the fecal-oral route and it is very virulent. Without complications, symptoms last about 5-10 days in general.
In the laboratory, stool samples are tested for the toxin via a rapid immunoassay ELISA test. The organism can also be identified by the MALDI-TOF and on the Biofire micro filmarray PCR in about an hour. This is a reportable organism to the state Public Health Laboratory, where it is serotyped and recorded, and then sent to other agencies that test and track cases and outbreaks.
In the laboratory, stool samples are tested for the toxin via a rapid immunoassay ELISA test. The organism can also be identified by the MALDI-TOF and on the Biofire micro filmarray PCR in about an hour. This is a reportable organism to the state Public Health Laboratory, where it is serotyped and recorded, and then sent to other agencies that test and track cases and outbreaks.
cyclospora infection/outbreak:
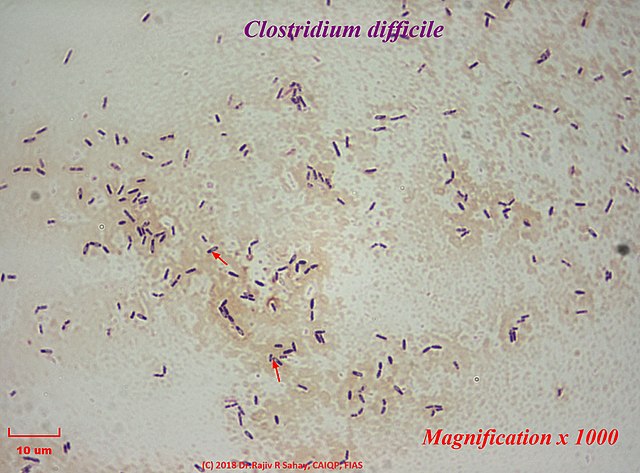
Clostridium difficile:
Clostridioides difficile (C. diff ) is a bacterium that causes life-threatening diarrhea, which can cause serious dehydration, pseudomembranous colitis, and can even be deadly. It is usually a consequence of taking antibiotics. "C. diff is a spore-forming, Gram-positive, anaerobic, rod-shaped bacillus that produces two exotoxins: toxin A and toxin B. It is a common cause of antibiotic-associated diarrhea (AAD) and accounts for 15 to 25% of all episodes of AAD." (CDC)
According to the CDC, those most at risk for this type of infection include:
Symptoms include:
If an individual has been taking antibiotics recently and develops symptoms of C. diff, they should be seen by a healthcare professional.
Prescribe and use antibiotics carefully. You can inquire about your patient’s past history of C. diff infection before prescribing." (CDC)
Other important interventions include:
"1. Isolate and initiate contact precautions for suspected or confirmed CDI
2. Confirm CDI in patients
3. Perform environmental cleaning to prevent CDILearn more about EPA List K agentsexternal icon
4. Develop infrastructure to support CDI prevention
5. Engage the facility antibiotic stewardship program
Supplemental interventions include:
b Patients with clinically significant unexplained diarrhea
c Infection control measures should be maintained for patients with EIA-positive test result as well as for those with EIA-negative but PCR-positive test result
d Non-infectious causes of diarrhea include inflammatory bowel disease, and therapies such as eternal tube feeding, intensive cancer chemotherapy, or laxatives.
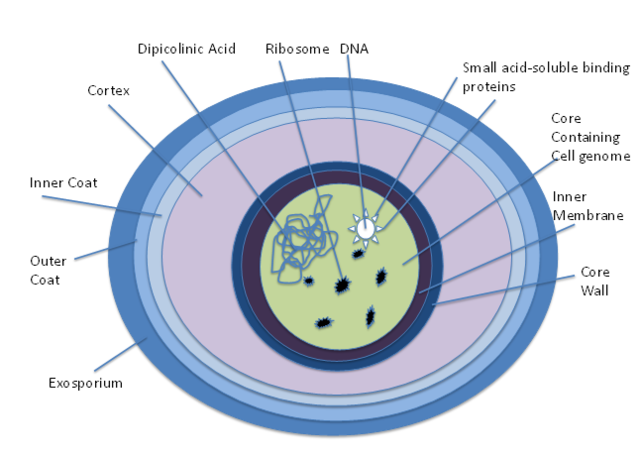
Endospores:
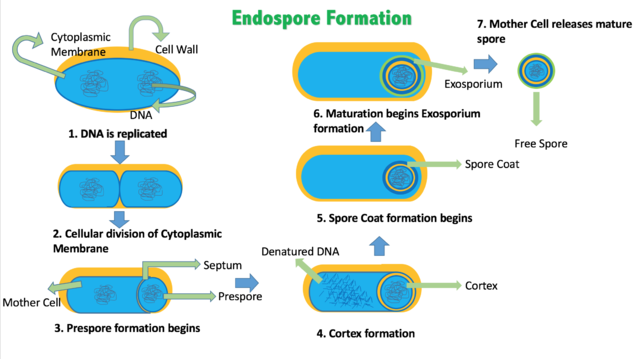
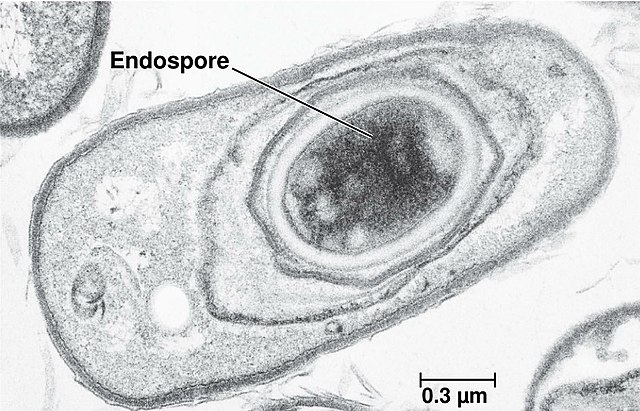
When C. diff germs are outside the body, they become spores, which can survive extreme conditions. The vegetative portion of the bacteria dies when antibiotics treat it or when disinfected, but in this case, sporulation causes endospores to form, which can stay in this state until conditions are favorable once again. These spores are an inactive form of the germ and have a thick protective coating allowing them to live for months or sometimes years on surfaces and in the soil. The germs regenerate again when these spores are swallowed and reach the intestines, where conditions are favorable. Healthy people will often not be infected even if the spores reach their intestines, but if your immune system is weakened or you’ve recently taken antibiotics, you could get sick. The thing is, alcohol-based hand rubs DO NOT kill or destroy the endospores. The best means of destroying this bug is following hand hygiene by washing the hands thoroughly with soap and water, lather it up, scrub for at least 20 seconds, between the fingers, front and back of the hands and wrists, between the grooves, under the nails, rinse, and pat dry with a clean paper towel.
Lab tests:
Lab tests include PCR, such as the GenEXPERT cartridge seen below, rapid antigen detection for C. diff: These are rapid tests (<1 hour) that detect the presence of C. diff antigen glutamate dehydrogenase (GDH). Because results of antigen testing alone are nonspecific, antigen assays have been employed in combination with tests for toxin detection, PCR, or toxigenic culture in two-step testing algorithms. Other tests include:
"Although in about 20% of patients, CDI will resolve within two to three days of discontinuing the antibiotic to which the patient was previously exposed, CDI should usually be treated with an appropriate course (about 10 days) of treatment, including oral vancomycin or fidaxomicin. After treatment, repeat C. diff testing is not recommended if the patient’s symptoms have resolved, as patients often remain colonized." (CDC)
What are the steps to prevent spread?
"If a patient has had ≥ 3 stools in 24 hours:
How can CDI be prevented in hospitals and other healthcare settings?
What can I use to clean and disinfect surfaces and devices potentially contaminated with C. diff?
"Surfaces should be kept clean, and body substance spills should be managed promptly, as outlined in CDC’s Guidelines for Environmental Infection Control in Health-Care Facilities. Routine cleaning should be performed prior to disinfection. EPA-registered disinfectants with a sporicidal claim have been used with success for environmental surface disinfection in those patient-care areas where surveillance and epidemiology indicate ongoing transmission of C. diff.
Note: EPA-registered disinfectants (List K) are recommended for use in patient-care areas. When choosing a disinfectant, check product labels for inactivation claims, indications for use, and instructions." (CDC)
According to the CDC, those most at risk for this type of infection include:
- "People 65 and older who take antibiotics and receive medical care
- People staying in hospitals and nursing homes for a long period of time
- People with weakened immune systems or previous infection with C. difficile
- Previous infection"
- "One in 6 people who’ve had C. diff will get it again in the subsequent 2-8 weeks.
- If you start having symptoms again, seek medical care.
- For those with repeat infections, innovative treatments, including fecal microbiota transplants, have shown promising results."
Symptoms include:
- malodorous diarrhea: loose, watery stools (poop) for several days, multiple times a day (smells like cow manure)
- fever
- abdominal pain and tenderness
- bloating
- loss of appetite
- nausea
- fatigue
- inflammation of the colon (colitis)
- toxic megacolon
- perforations of the colon
- sepsis
- can cause death if untreated
If an individual has been taking antibiotics recently and develops symptoms of C. diff, they should be seen by a healthcare professional.
- "Developing diarrhea is fairly common while on, or after taking, antibiotics, but in only a few cases will that diarrhea be caused by C. diff. If your diarrhea is severe, do not delay getting medical care.
- Your healthcare professional will review your symptoms and order a lab test of a stool (poop) sample.
- If the test is positive, you’ll take a specific antibiotic (e.g. vancomycin or fidaxomicin) for at least 10 days. If you were already taking an antibiotic for another infection, your healthcare professional might ask you to stop taking it if they think it’s safe to do so.
- Your healthcare team might decide to admit you to the hospital, in which case they will use enteric contact precautions, such as wearing gowns and gloves, to prevent the spread of C. diff to themselves and to other patients." (CDC)
- Washing one's hands with soap and water every time you use the bathroom and always before you eat.
- Try to use a separate bathroom if you have diarrhea.
- Take showers and wash with soap.
Prescribe and use antibiotics carefully. You can inquire about your patient’s past history of C. diff infection before prescribing." (CDC)
Other important interventions include:
- 'assessing patients for CDI risk factors
- timely testing of patients with compatible signs and symptoms
- using contact isolation precautions, including wearing gloves and a gown for patients with unexplained diarrhea
- following hand hygiene practices, including before seeing a patient and after removal of gloves
- daily and terminal cleaning of patient rooms" (CDC)
"1. Isolate and initiate contact precautions for suspected or confirmed CDI
- Create nurse-driven protocols (a) to facilitate rapid isolation of patients with suspected or confirmed CDI
- Patients with diarrhea should be isolated while evaluation for the cause is ongoing (e.g., patient remains isolated during a trial off laxatives)
- For suspected patients, ensure rapid evaluation by healthcare personnel and infection prevention
- Place symptomatic patients b on contact precautions, in a single-patient room with a dedicated toilet
- If single-patient rooms are not available, room patients with confirmed CDI together
- For patients with confirmed CDI, maintain contact precautions for at least 48 hours after diarrhea has resolved, or longer, up to the duration of hospitalizationc
- Adhere to recommended hand hygiene practices
- Use dedicated patient-care equipment (e.g., blood pressure cuffs, stethoscopes)
- Implement daily patient bathing or showering with soap and water
- When transferring patients, notify receiving wards or facilities about the patient’s CDI status so contact precautions are maintained at the patient’s new location
2. Confirm CDI in patients
- Clinical personnel
- Assess for appropriateness of testing: Consider other infectious or non-infectiousd causes of diarrhea before testing for CDI
- Discontinue laxatives and wait for at least 48 hours before testing if still symptomatic
- Once a patient has a positive CDI test do not repeat testing to detect cure; tests may remain positive for ≥6 weeks
- Laboratory personnel
- Implement laboratory procedures to ensure testing of only appropriate specimens (e.g., unformed stool) for C. difficile or its toxins
- For sites where appropriateness of testing is an issue, consider implementing two-step testing (e.g., high sensitivity NAAT or GDH test followed by high-specificity toxin test, rather than NAAT alone) to improve diagnostic accuracy
- Report test results immediately to clinical care providers and infection control personnel through reliable means (e.g., a laboratory alert system)
- Implement laboratory procedures to ensure testing of only appropriate specimens (e.g., unformed stool) for C. difficile or its toxins
3. Perform environmental cleaning to prevent CDILearn more about EPA List K agentsexternal icon
- Create daily and terminal cleaning protocols and checklists for patient-care areas and equipment
- Perform daily cleaning of CDI patient rooms using a C. difficile sporicidal agent (EPA List K agent)
- Clean and disinfect the patient-care environment (including the immediate vicinity around a CDI patient and high touch surfaces) at least once a day, including toilets
- Clean and disinfect all shared equipment prior to use with another patient (e.g., wheelchairs, gurneys)
- Perform terminal cleaning after CDI patient transfer/discharge with a C. difficile sporicidal agent (EPA List K agent)
- Clean additional areas that are contaminated during transient visits by patients with suspected or confirmed CDI (e.g., Radiology, Emergency Departments, Physical Therapy) with a C. difficile sporicidal agent (EPA List K agent)
4. Develop infrastructure to support CDI prevention
- Incorporate reduction of CDI into the facility healthcare-associated infection prevention program, including but not limited to the design, implementation, evaluation, and feedback of intervention results
- Include a multidisciplinary workgroup, including physicians, nursing, environmental services, and antibiotic stewardship to identify and implement the below strategies and to use data for action
- Monitor facility CDI rates, and target units with highest incidence of CDI for evaluation and intervention
- Review hospital-onset CDI cases to help identify potential gaps and opportunities for improvement
- Review should focus on opportunities for improvement across each strategy (e.g., test indications, antibiotic appropriateness)
- Utilize findings to engage relevant care teams and staff in gap remediation and performance improvement as soon after the CDI case as possible
- Educate and train healthcare personnel on prevention practices for CDI
- Routinely audit
- Adherence to hand hygiene and contact precautions
- Adequacy of room cleaning using methods described in “Options for Evaluating Environments Cleaning”
- Provide CDI rates and other performance improvement measures to senior leadership, clinical providers, laboratory personnel, environmental services, and other stakeholders
- Notify appropriate individuals and facility departments about changes in the incidence (or frequency), complications (including recurrences), or severity of CDI
5. Engage the facility antibiotic stewardship program
- Implement the 7 Core Elements of Hospital Antibiotic Stewardship
- Assess the appropriateness of prescribing antibiotics that pose the highest risk for CDI, especially fluoroquinolones, carbapenems, and 3rd and 4th generation cephalosporins
- Develop facility-specific treatment recommendations for common infections that include first- and second-line antibiotics
- Evaluate antibiotic treatment of conditions that commonly lead to high-risk antibiotic use, such as asymptomatic bacteriuria and common infections such as urinary tract infection and community-acquired pneumonia, to minimize the use of high-risk antibiotics
- Ensure that patients receive the shortest effective duration of antibiotic therapy
- Include inpatient antibiotic duration when determining post-discharge antibiotic duration
Supplemental interventions include:
- Dedicate healthcare personnel to the care of patients with CDI only (i.e., without responsibility to care for non-CDI patients), who are typically cohorted on a single ward or unit, to minimize the risk of transmission to others
- Restrict the use of antibiotics with the highest risk for CDI (e.g., fluoroquinolones, carbapenems, 3rd and 4th generation cephalosporins)
- Limit the use of other medications (e.g., proton pump inhibitors) that are hypothesized to increase risk for CDI
- Evaluate and test asymptomatic patients at high risk for CDI to detect carriage
- Isolate patients that test positive, but do not treat in the absence of symptoms
- Consider isolating patients with carriage and diarrhea, until the diarrhea resolves, as a routine strategy
- Use of additional disinfection of CDI patient rooms with no-touch technologies (e.g., UV light)
- Expand the use of environmental disinfection strategies (e.g., sporicidal agents [EPA List K agent]) for daily and terminal cleaning in all rooms on affected units
- Participate in regional CDI prevention activities"
b Patients with clinically significant unexplained diarrhea
c Infection control measures should be maintained for patients with EIA-positive test result as well as for those with EIA-negative but PCR-positive test result
d Non-infectious causes of diarrhea include inflammatory bowel disease, and therapies such as eternal tube feeding, intensive cancer chemotherapy, or laxatives.
Endospores:
When C. diff germs are outside the body, they become spores, which can survive extreme conditions. The vegetative portion of the bacteria dies when antibiotics treat it or when disinfected, but in this case, sporulation causes endospores to form, which can stay in this state until conditions are favorable once again. These spores are an inactive form of the germ and have a thick protective coating allowing them to live for months or sometimes years on surfaces and in the soil. The germs regenerate again when these spores are swallowed and reach the intestines, where conditions are favorable. Healthy people will often not be infected even if the spores reach their intestines, but if your immune system is weakened or you’ve recently taken antibiotics, you could get sick. The thing is, alcohol-based hand rubs DO NOT kill or destroy the endospores. The best means of destroying this bug is following hand hygiene by washing the hands thoroughly with soap and water, lather it up, scrub for at least 20 seconds, between the fingers, front and back of the hands and wrists, between the grooves, under the nails, rinse, and pat dry with a clean paper towel.
Lab tests:
Lab tests include PCR, such as the GenEXPERT cartridge seen below, rapid antigen detection for C. diff: These are rapid tests (<1 hour) that detect the presence of C. diff antigen glutamate dehydrogenase (GDH). Because results of antigen testing alone are nonspecific, antigen assays have been employed in combination with tests for toxin detection, PCR, or toxigenic culture in two-step testing algorithms. Other tests include:
- "Toxin testing for C. diff:
- Tissue culture cytotoxicity assay detects toxin B only. This assay requires technical expertise to perform, is costly, and requires 24 to 48 hours for a final result. It does provide specific and sensitive results for CDI. While it served as a historical gold standard for diagnosing clinically significant disease caused by C. diff, it is recognized as less sensitive than PCR or toxigenic culture for detecting the organism in patients with diarrhea.
- Enzyme immunoassay detects toxin A, toxin B, or both A and B. Due to concerns over toxin A-negative, B-positive strains causing disease, most laboratories employ a toxin B-only or A and B assay. Because these are same-day assays that are relatively inexpensive and easy to perform, they are popular with clinical laboratories. However, there are increasing concerns about their relative insensitivity (less than tissue culture cytotoxicity and much less than PCR or toxigenic culture).
- C. diff toxin is very unstable. The toxin degrades at room temperature and might be undetectable within two hours after collection of a stool specimen. False-negative results occur when specimens are not promptly tested or kept refrigerated until testing can be done.
- Stool culture for C. diff: While this is the most sensitive test available, it is the one most often associated with false-positive results due to the presence of nontoxigenic C. diff strains. However, this can be overcome by testing isolates for toxin production (i.e. so-called “toxigenic culture”). Nonetheless, stool cultures for C. diff are labor-intensive, require an appropriate culture environment to grow anaerobic microorganisms, and have a relatively slow turnaround time (i.e. results available in 48 to 96 hours), making them less clinically useful overall. Results of toxigenic cultures do serve as a gold standard against which other test modalities are compared in clinical trials of performance." (CDC)
"Although in about 20% of patients, CDI will resolve within two to three days of discontinuing the antibiotic to which the patient was previously exposed, CDI should usually be treated with an appropriate course (about 10 days) of treatment, including oral vancomycin or fidaxomicin. After treatment, repeat C. diff testing is not recommended if the patient’s symptoms have resolved, as patients often remain colonized." (CDC)
What are the steps to prevent spread?
"If a patient has had ≥ 3 stools in 24 hours:
- Order a C. diff test if other etiologies of diarrhea (e.g., stool softener or laxative use) are considered unlikely.
- In patient being evaluated for C. diff, reassess appropriateness of antibiotics.
- Wear gloves and a gown when treating patients with C. diff, even during short visits. Gloves are important because hand sanitizer doesn’t kill C. diff and handwashing might not be sufficient alone to eliminate all C. diff spores.
- Isolate patients with possible C. diff immediately, even if you only suspect CDI.
- Continue isolation and contact precautions.
- When a patient transfers, notify the new facility if the patient has or had a C. diff infection. (Inter-Facility Infection Control Transfer Form pdf icon[PDF – 3 pages])
- Clean room surfaces thoroughly on a daily basis while treating a patient with C. diff and upon patient discharge or transfer using an EPA-approved spore-killing disinfectant.
- Use antibiotics appropriately." (CDC)
How can CDI be prevented in hospitals and other healthcare settings?
- "Improving Diagnosis and Management
- Use contact precautions for patients with known or suspected CDI:
- Place these patients in private rooms. If private rooms are not available, they can be placed in rooms (cohorted) with other CDI patients.
- Wear gloves and a gown when entering CDI patient rooms and during their care.
- As no single method of hand hygiene will eliminate all C. diff spores, using gloves to prevent hand contamination remains the cornerstone for preventing C. diff transmission via the hands of healthcare personnel.
- Always perform hand hygiene after removing gloves.
- Clinical Practice Guidelines for Clostridium difficile Infection in Adults and Children: 2017
Use antibiotics appropriately. - If your institution experiences an outbreak, consider using soap and water instead of alcohol-based hand sanitizers for hand hygiene after removing gloves while caring for patients with CDI.
- Dedicate or perform cleaning and disinfection of any shared medical equipment between patients.
- Continue CDI precautions at least until diarrhea ceases.
- Because CDI patients continue to shed the organism for a number of days following cessation of diarrhea, some institutions routinely continue isolation and contact precautions for either several days beyond symptom resolution or until discharge, depending upon the type of setting and average length of stay.
- Implement an environmental cleaning and disinfection strategy.
- Ensure adequate cleaning and disinfection of environmental surfaces and reusable devices, especially items likely to be contaminated with feces and surfaces that are touched frequently.
- Ensure daily and terminal cleaning of patient rooms.
- Use an Environmental Protection Agency (EPA)-registered disinfectant with a sporicidal claim for environmental surface disinfection after cleaning in accordance with label instructions. (Note: Only hospital surface disinfectants listed on EPA’s List K are registered as effective against C. diff spores).
- Follow the manufacturer’s instructions for disinfection of endoscopes and other devices." (CDC)
- Ensure adequate cleaning and disinfection of environmental surfaces and reusable devices, especially items likely to be contaminated with feces and surfaces that are touched frequently.
- Use contact precautions for patients with known or suspected CDI:
What can I use to clean and disinfect surfaces and devices potentially contaminated with C. diff?
"Surfaces should be kept clean, and body substance spills should be managed promptly, as outlined in CDC’s Guidelines for Environmental Infection Control in Health-Care Facilities. Routine cleaning should be performed prior to disinfection. EPA-registered disinfectants with a sporicidal claim have been used with success for environmental surface disinfection in those patient-care areas where surveillance and epidemiology indicate ongoing transmission of C. diff.
Note: EPA-registered disinfectants (List K) are recommended for use in patient-care areas. When choosing a disinfectant, check product labels for inactivation claims, indications for use, and instructions." (CDC)
Norovirus:
Norovirus is the main cause of the "stomach bug" we are so familiar with, and the main cause of vomiting and diarrhea with stomach pain, fever, chills, body aches, as well as foodborne illness, in the USA. It causes acute gastroenteritis, and it spreads easily and rapidly and it is highly contagious. Hospitals, schools, the military, daycare centers, healthcare facilities are all at risk. Because there are so many strains, and because the virus mutates so rapidly, you can become sick with this virus many times throughout life. It affects all ages and everyone is at risk for infection, but the biggest risk of this virus is rapid dehydration, which can quickly become serious.
The onset of symptoms occurs within about 12-48 hours after exposure. Recovery takes about 1-3 days, but a person can still transmit it even after this and even once they are feeling better. Because this illness leads to multiple bouts of vomiting and diarrhea throughout the duration of the illness, dehydration is a risk that can result in decreased output of urine, extreme thirst, sunken skin and eyes, dry mouth and throat, dizziness when standing up, and extreme fatigue and fussiness in children.
This virus spreads via the fecal-oral route. If someone accidentally ingests a virus particle via stool, fecal matter or vomitus, it can cause infection. Sick individuals shed billions of norovirus particles when infected, and they can continue to transmit the virions for up to two weeks after they feel better. It spreads by sharing food, drink or utensils with someone who is ill, eating food handled by an infected individual who has not washed their hands, eating foods or drinking liquids contaminated with the virus, or touching inanimate objects (fomite) that are contaminated with virus particles. Septic tank leaks, washing produce or oysters with contaminated water, improper disinfection of water and surfaces, vomiting into a public toilet where particles fly onto other surfaces, getting vomited on, are just several ways that this virus can spread.
It is important to realized that alcohol-based hand sanitizer is ineffective against norovirus. It is one of the few viruses that is not killed by it. The most effective means of prevention is washing one's hands frequently with soap and water for at least 20 seconds before and after direct contact, before and after caring for a patient, before and after going to the bathroom, after shaking hands, after touching surfaces, before eating, preparing or handling food, before administering medication. Cooking shellfish or oysters to an internal temperature of at least 145 degrees C is also important for protection from foodborne norovirus. Sanitizing, disinfecting, and sterilizing equipment is also key to prevention.
Here are the CDC's guidelines:
Do not prepare and handle food or care for others when you are sick.
"You should not prepare food for others or provide healthcare while you are sick and for at least 2 days (48 hours) after symptoms stop. This also applies to sick workers in restaurants, schools, daycares, long-term care facilities, and other places where they may expose people to norovirus." (CDC)
Clean and disinfect surfaces:
After someone vomits or has diarrhea, always clean well and disinfect the entire area immediately.
Make household bleach solution: To disinfect, you should use a chlorine bleach solution with a concentration of 1,000 to 5,000 ppm (5 to 25 tablespoons of household bleach [5% to 8%] per gallon of water) or use an EPA-registered disinfecting product against norovirus.
You should:
- Wear rubber or disposable gloves and wipe the entire area with paper towels and throw them in a plastic trash bag.
- Disinfect the area as directed on the product label.
- Leave the bleach disinfectant on the affected area for at least 5 minutes
- Clean the entire area again with soap and hot water.
- Wash laundry, take out the trash, and wash your hands.
You should:
- Wear rubber or disposable gloves.
- Handle items carefully without shaking them.
- Wash the items with detergent and hot water at the maximum available cycle length and then machine dry them at the highest heat setting.
- Wash your hands after with soap and water." (CDC)
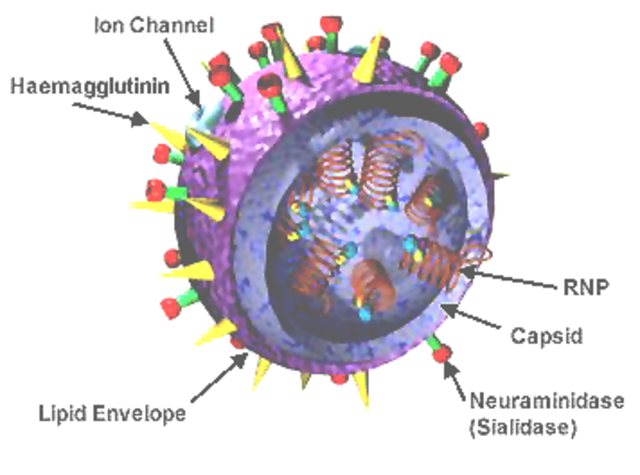
Influenza A & B:
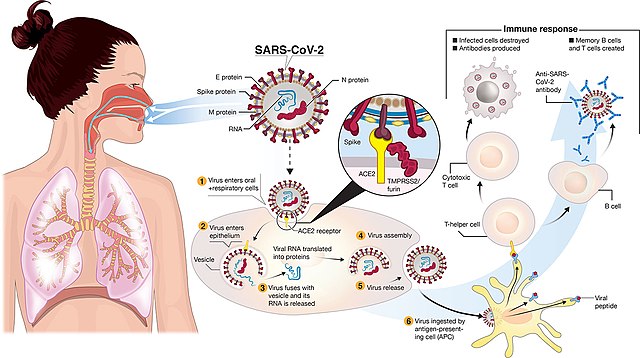
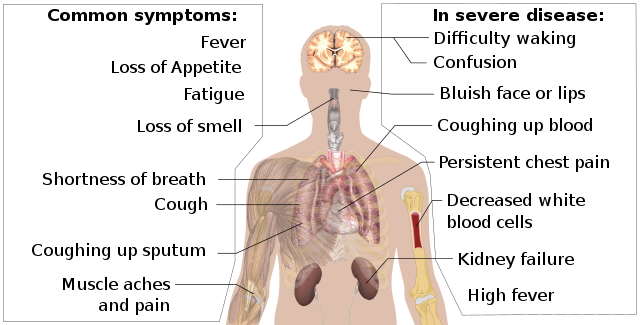
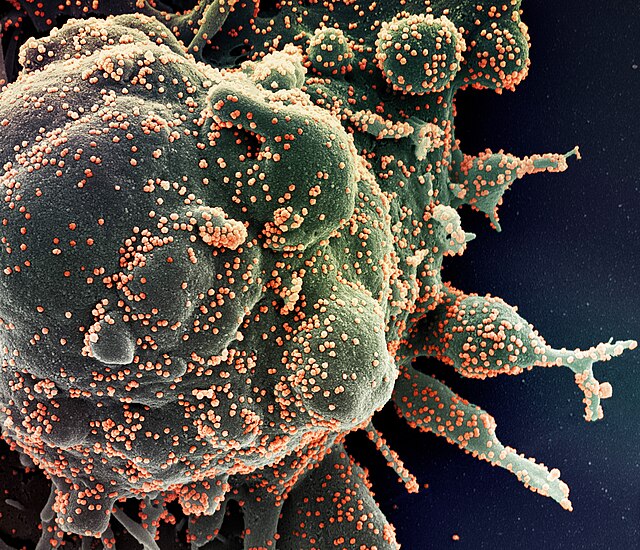
SARS-COVID-19:
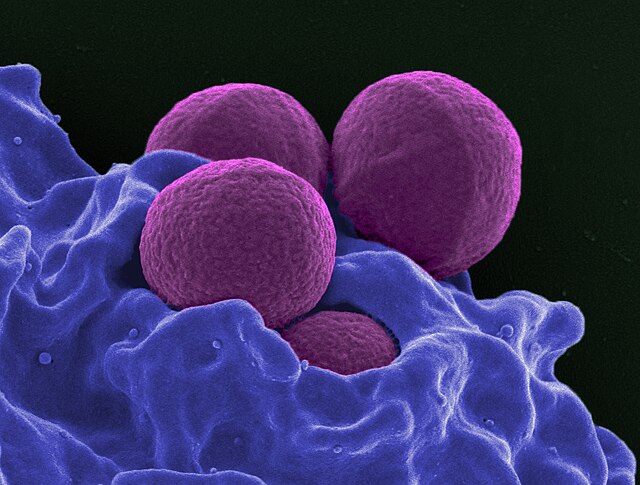
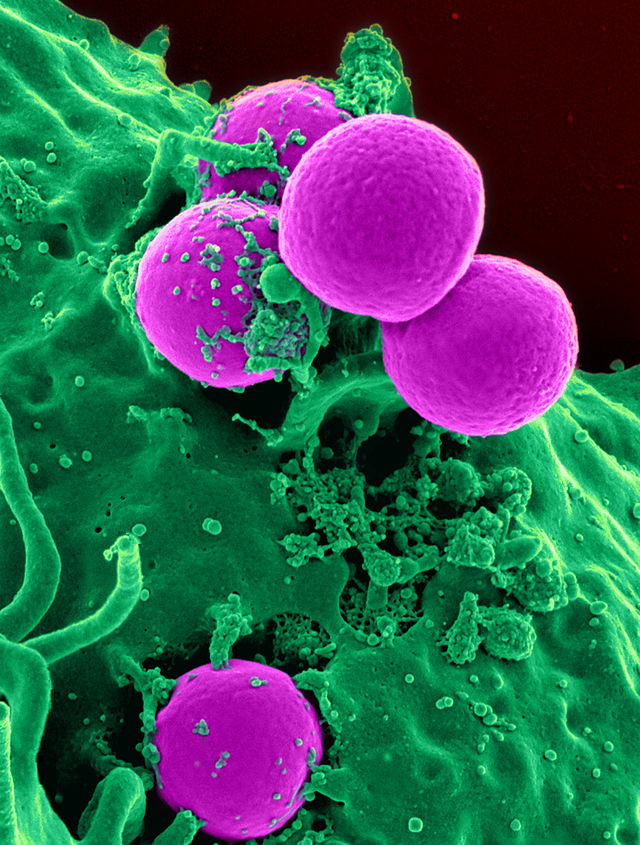
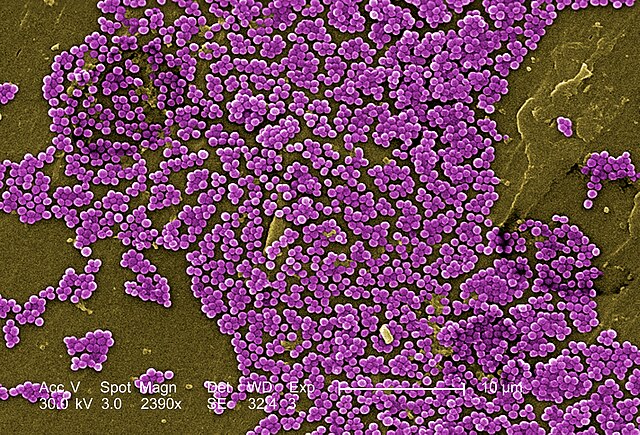
MRSA:
MRSA skin infections often appear as wounds or boils that are
"The symptoms of a MRSA infection depend on the part of the body that is infected." (CDC) For example, people with MRSA skin infections often can get swelling, warmth, redness, and pain in infected skin. An infection cannot be confirmed whether it is due to MRSA or another type of bacteria without laboratory tests ordered by the doctor. Most S. aureus skin infections, including MRSA, appear as a bump or infected area on the skin that might be:
Anyone is at risk for MRSA, however, the risk increases with activities or places where people are close together or crowded, where skin-to-skin contact is experienced, and if there is shared equipment or supplies, especially if they are improperly cleaned between use. Some of the people who carry MRSA (this is called "colonization") can develop a MRSA infection if conditions are just right and the bacteria gets into places in the body, causing an opportunistic infection. Non-intact skin, when the skin barrier is compromised, such as when there are abrasions or incisions, is often the site of an opportunistic MRSA infection. Athletes in gyms regularly, daycares, schools, military personnel in barracks, and inpatients, surgery patients, or medical devices inserted inside the body place a person at higher risk of MRSA infection.
MRSA can be spread in the community setting by coming into contact with infected people or inanimate objects (fomites) that are carrying the bacteria. This includes through direct contact with a contaminated wound or by sharing personal items, such as towels or razors, that have touched infected skin. This is why it is so important for gyms, swimming pools, saunas, spas, salons, changing rooms, surgical suites, hospital rooms, etc... to be thoroughly cleaned and disinfected. Drug use is also linked to MRSA infection and spread. In fact, according to the CDC, "people who inject drugs are 16 times more likely to develop a serious staph infection." (CDC) Also, according to the CDC, approximately 5% of patients in U.S. hospitals carry MRSA in their nose or on their skin." (CDC) For this reason, this is why many hospital infection control programs have surveillance policies to swab and test patients who are at risk to see if they are colonized with and carrying MRSA.
In the healthcare setting, MRSA is often spread by direct contact with an infected wound or from contaminated hands, usually those of healthcare providers. Also, people who carry MRSA but do not have signs of infection can spread the bacteria to others (i.e., people who are colonized). This is why it is so important for healthcare workers to follow the infection control measures to put patients on contact isolation precautions, follow the protocol carefully, make sure a sign is on the door of a hospital inpatient, in order to make sure that MRSA does not spread or cause infection in a susceptible individual.
We can take measures to prevent the spread of MRSA and reduce the risk of infection:
Because we cannot tell simply by looking at the skin if it’s a staph infection (including MRSA),
seeking medical care early makes it less likely that the infection will become serious.
If you or someone in your family experiences the signs and symptoms of MRSA:
"The CDC is actively participating in several short- and long-term MRSA surveillance (infection tracking) projects that involve collaboration with health departments, individual hospitals, and academic medical centers, among others. Understanding the burden of MRSA infections—how much is occurring, where it is happening, and how it is being spread – is essential for developing effective prevention programs and measuring their impact." (CDC)
Also, according to the CDC, "studies show that about one in three (33%) people carry S. aureus bacteria in their nose, usually without any illness. About two in every 100 people carry MRSA. Although many people carry MRSA bacteria in their nose, most do not develop serious MRSA infections." (CDC) This is why surveillance swabbing and testing, often by PCR, is key, because it can detect the mecA gene associated with MRSA.
Encouraging progress was made to decrease MRSA bloodstream infections in healthcare from 2005-2012, when the rates of MRSA bloodstream infections dropped by 17.1% each year during that timeframe (CDC). However it was also noted that declines in MRSA bloodstream infections slowed from 2013-2016, when no significant change was detected. (CDC)
"Information about MRSA rates in hospitals across the United States is available through the National Healthcare Safety Network (NHSN), the nation’s most widely used healthcare-associated infection tracking system." (CDC)
Since prevention is key, many studies, including a 2019 publication about Veterans Affairs Medical Centers, demonstrate that MRSA prevention efforts can reduce infections, and this is part of a hospital or clinic's infection prevention program. In fact, successful MRSA prevention requires action both at the healthcare facility level, among healthcare providers and healthcare leadership, working together and meeting and discussing programs, progress, clinical education, and ways to improve. Healthcare facilities can make prevention of MRSA infections a priority, assess their relevant data, implement prevention actions and evaluate progress.
Healthcare providers can follow current prevention recommendations for device- and procedure- related infections, treat infections appropriately and rapidly if they do occur, and educate patients about ways to avoid infection and spread, and about early signs of sepsis. Infection control practices should be reinforced on an ongoing basis, including the use of competency-based training and monitoring of adherence with feedback of results for practices including hand hygiene, environmental cleaning and disinfection, and use of personal protective equipment (PPE).
According to the CDC, "core and supplemental strategies should be taken into consideration. Core strategies are supported by published evidence; supplemental strategies are generally supported by less evidence and could be considered for use when reduction goals are not met after implementation of core interventions or when facilities need to implement a more aggressive prevention strategy." (CDC)
"The first step in developing a HO SA BSI prevention strategy is to review recent episodes of HO SA BSI to identify common risk factors and underlying syndromes that can help identify the populations to prioritize. Elements that could be reviewed include associated syndromes (e.g., wound infections or pneumonia) that may have led to the BSI, unit types, presence of indwelling devices such as central venous catheters (CVCs), and prior invasive procedures or surgeries. Based on this review of facility-level data, the most impactful core and supplemental strategies can be chosen." (CDC)
1. IMPLEMENT INTERVENTIONS TO REDUCE DEVICE AND PROCEDURE RELATED HEALTHCARE-ASSOCIATED INFECTIONS
2. IMPLEMENT DECOLONIZATION AND PATHOGEN REDUCTION STRATEGIES FOR HIGH RISK PATIENTS DURING HIGH RISK PERIODS
3. IMPLEMENT INTERVENTIONS TO PREVENT TRANSMISSION OF METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS (MRSA) IN ACUTE CARE
4. DEVELOP INFRASTRUCTURE TO SUPPORT HO SA BSI PREVENTION
Table 1: Summary of Decolonization and Pathogen Reduction Strategies by Central Venous Catheter (CVC) or Midline Catheter Presence and Unit Type:
Patient Type Intensive Care Unit Non-Intensive Care Unit
CVC or Midline Catheter Present Topical chlorhexidine gluconate (at least 2%) + Intranasal antistaphylococcal antibiotic/antiseptic (e.g. mupirocin or iodophor) (core strategy) Topical chlorhexidine gluconate (at least 2%) + Intranasal antistaphylococcal antibiotic/antiseptic (e.g. mupirocin or iodophor) ( supplemental strategy)
No CVC or Midline Catheter Topical chlorhexidine gluconate (at least 2%) + Intranasal antistaphylococcal antibiotic/antiseptic (i.e. mupirocin or iodophor) (core strategy)
None (note that decolonization or pathogen reduction strategies may apply to pre-operative surgical patients outside the intensive care unit- see section 2) (CDC)
Precautions: Contact Precautions"CDC recommends the use of Contact Precautions in inpatient acute care settings for patients known to be colonized or infected with epidemiologically important multidrug-resistant organisms (MDROs) including MRSA.
From 2005 to 2014, the overall estimated incidence of invasive MRSA infections from normally sterile sites (i.e., blood, pleural fluid, etc.) in the United States declined by 40% and the estimated incidence of invasive hospital-onset MRSA infections declined by 65%. Interventions designed to decrease risk of device and procedure-associated infections and interventions to reduce transmission, like Contact Precautions and hand hygiene, both likely contributed to these decreases. The relative contribution of Contact Precautions in comparison to other interventions is unknown.
In the past 5 years, more than a dozen studies, review articles, and editorials have sought to address whether Contact Precautions should continue to be recommended for endemic MDROs like MRSA. The impact of discontinuing Contact Precautions for MRSA-colonized or infected patients has been assessed primarily in single-center studies using lower quality quasi-experimental designs that have not identified changes in MRSA infection or acquisition rates. These studies likely underestimate the impact of discontinuing Contact Precautions, including the effect on downstream adverse events (e.g., post-discharge infections).
Based on the current evidence, CDC continues to recommend the use of Contact Precautions for MRSA-colonized or infected patients. CDC will continue to evaluate the evidence on Contact Precautions as it becomes available. In addition, CDC continues to work with partners to identify and evaluate other measures to decrease transmission of MDROs in healthcare settings." (CDC)
"MRSA remains an important healthcare pathogen and the prevention of MRSA infections is a priority for CDC. CDC estimates that MRSA is responsible for more than 70,000 severe infections and 9,000 deaths per year.
Make preventing staph infections (MRSA and MSSA) a priority in healthcare facilities:
- red (inflamed)
- swollen (edema)
- painful, or
- have pus or other drainage (dead neutrophils, cellular debris, bacteria)
"The symptoms of a MRSA infection depend on the part of the body that is infected." (CDC) For example, people with MRSA skin infections often can get swelling, warmth, redness, and pain in infected skin. An infection cannot be confirmed whether it is due to MRSA or another type of bacteria without laboratory tests ordered by the doctor. Most S. aureus skin infections, including MRSA, appear as a bump or infected area on the skin that might be:
- red
- swollen
- painful
- warm to the touch
- full of pus or other drainage
- accompanied by a fever
Anyone is at risk for MRSA, however, the risk increases with activities or places where people are close together or crowded, where skin-to-skin contact is experienced, and if there is shared equipment or supplies, especially if they are improperly cleaned between use. Some of the people who carry MRSA (this is called "colonization") can develop a MRSA infection if conditions are just right and the bacteria gets into places in the body, causing an opportunistic infection. Non-intact skin, when the skin barrier is compromised, such as when there are abrasions or incisions, is often the site of an opportunistic MRSA infection. Athletes in gyms regularly, daycares, schools, military personnel in barracks, and inpatients, surgery patients, or medical devices inserted inside the body place a person at higher risk of MRSA infection.
MRSA can be spread in the community setting by coming into contact with infected people or inanimate objects (fomites) that are carrying the bacteria. This includes through direct contact with a contaminated wound or by sharing personal items, such as towels or razors, that have touched infected skin. This is why it is so important for gyms, swimming pools, saunas, spas, salons, changing rooms, surgical suites, hospital rooms, etc... to be thoroughly cleaned and disinfected. Drug use is also linked to MRSA infection and spread. In fact, according to the CDC, "people who inject drugs are 16 times more likely to develop a serious staph infection." (CDC) Also, according to the CDC, approximately 5% of patients in U.S. hospitals carry MRSA in their nose or on their skin." (CDC) For this reason, this is why many hospital infection control programs have surveillance policies to swab and test patients who are at risk to see if they are colonized with and carrying MRSA.
In the healthcare setting, MRSA is often spread by direct contact with an infected wound or from contaminated hands, usually those of healthcare providers. Also, people who carry MRSA but do not have signs of infection can spread the bacteria to others (i.e., people who are colonized). This is why it is so important for healthcare workers to follow the infection control measures to put patients on contact isolation precautions, follow the protocol carefully, make sure a sign is on the door of a hospital inpatient, in order to make sure that MRSA does not spread or cause infection in a susceptible individual.
We can take measures to prevent the spread of MRSA and reduce the risk of infection:
- "Maintain good hand and body hygiene. Clean hands often, and clean your body regularly, especially after exercise. Keeping your hands clean is one of the most important steps you can take to avoid getting sick and spreading germs like MRSA. Use soap and water to clean your hands if available. After wetting your hands and adding soap, scrub your hands for at least 20 seconds. If you cannot access soap and water, use an alcohol-based hand sanitizer that contains at least 60% alcohol to clean hands. Apply the sanitizer to one hand, rub your hands together, trying to cover all surfaces of your hands and fingers until your hands are dry.
- Keep cuts, scrapes, and wounds clean and covered until healed.
- Avoid sharing personal items such as towels and razors. Methicillin-resistant Staphylococcus aureus (MRSA) can survive on some surfaces, like towels, razors, furniture, and athletic equipment for hours, days, or even weeks. It can spread to people who touch a contaminated surface, and MRSA can cause infections if it gets into a cut, scrape, or open wound.
- Cleaners or detergents are products that remove soil, dirt, dust, organic matter, and germs (like bacteria, viruses, and fungi). They lift dirt and germs off surfaces so they can be rinsed away with water. Cleaning with a detergent is necessary to remove dirt that can prevent disinfectants from working. Some disinfectants have a cleaning agent mixed in, check the label to know which product you have.
- Disinfectants are chemical products that are used to kill germs in healthcare settings. Disinfectants effective against Staphylococcus aureus, or staph, are also effective against MRSA. The disinfectant’s label should have a list of germs that the product can kill, along with an Environmental Protection Agency (EPA) registration number. These products are also sold at grocery and other retail stores and may be helpful when someone has an infected wound.
- When cleaning and disinfecting, focus on surfaces that frequently contact people’s bare skin like desks, chairs, benches, gym equipment, lockers, faucets, light switches and remote controls. In particular, clean any surfaces that could come into contact with uncovered wounds, cuts, or boils. In addition to cleaning surfaces, frequently cleaning hands and keeping wounds covered keeps MRSA from spreading.
Large surfaces, such as floors and walls, have not been associated with the spread of staph and MRSA. There is no evidence that spraying or fogging rooms or surfaces with disinfectants will prevent MRSA infections more effectively than the targeted approach of cleaning frequently touched surfaces and surfaces that have been exposed to open wounds. - Get care early if you think you might have an infection." (CDC)
Because we cannot tell simply by looking at the skin if it’s a staph infection (including MRSA),
seeking medical care early makes it less likely that the infection will become serious.
If you or someone in your family experiences the signs and symptoms of MRSA:
- "Contact your healthcare provider, especially if the symptoms are accompanied by a fever.
- Do not pick at or pop the sore.
- Cover the area with clean, dry bandages until you can see a healthcare provider.
- Clean your hands often." (CDC)
"The CDC is actively participating in several short- and long-term MRSA surveillance (infection tracking) projects that involve collaboration with health departments, individual hospitals, and academic medical centers, among others. Understanding the burden of MRSA infections—how much is occurring, where it is happening, and how it is being spread – is essential for developing effective prevention programs and measuring their impact." (CDC)
Also, according to the CDC, "studies show that about one in three (33%) people carry S. aureus bacteria in their nose, usually without any illness. About two in every 100 people carry MRSA. Although many people carry MRSA bacteria in their nose, most do not develop serious MRSA infections." (CDC) This is why surveillance swabbing and testing, often by PCR, is key, because it can detect the mecA gene associated with MRSA.
Encouraging progress was made to decrease MRSA bloodstream infections in healthcare from 2005-2012, when the rates of MRSA bloodstream infections dropped by 17.1% each year during that timeframe (CDC). However it was also noted that declines in MRSA bloodstream infections slowed from 2013-2016, when no significant change was detected. (CDC)
"Information about MRSA rates in hospitals across the United States is available through the National Healthcare Safety Network (NHSN), the nation’s most widely used healthcare-associated infection tracking system." (CDC)
Since prevention is key, many studies, including a 2019 publication about Veterans Affairs Medical Centers, demonstrate that MRSA prevention efforts can reduce infections, and this is part of a hospital or clinic's infection prevention program. In fact, successful MRSA prevention requires action both at the healthcare facility level, among healthcare providers and healthcare leadership, working together and meeting and discussing programs, progress, clinical education, and ways to improve. Healthcare facilities can make prevention of MRSA infections a priority, assess their relevant data, implement prevention actions and evaluate progress.
Healthcare providers can follow current prevention recommendations for device- and procedure- related infections, treat infections appropriately and rapidly if they do occur, and educate patients about ways to avoid infection and spread, and about early signs of sepsis. Infection control practices should be reinforced on an ongoing basis, including the use of competency-based training and monitoring of adherence with feedback of results for practices including hand hygiene, environmental cleaning and disinfection, and use of personal protective equipment (PPE).
According to the CDC, "core and supplemental strategies should be taken into consideration. Core strategies are supported by published evidence; supplemental strategies are generally supported by less evidence and could be considered for use when reduction goals are not met after implementation of core interventions or when facilities need to implement a more aggressive prevention strategy." (CDC)
"The first step in developing a HO SA BSI prevention strategy is to review recent episodes of HO SA BSI to identify common risk factors and underlying syndromes that can help identify the populations to prioritize. Elements that could be reviewed include associated syndromes (e.g., wound infections or pneumonia) that may have led to the BSI, unit types, presence of indwelling devices such as central venous catheters (CVCs), and prior invasive procedures or surgeries. Based on this review of facility-level data, the most impactful core and supplemental strategies can be chosen." (CDC)
1. IMPLEMENT INTERVENTIONS TO REDUCE DEVICE AND PROCEDURE RELATED HEALTHCARE-ASSOCIATED INFECTIONS
- Central line-associated bloodstream infection (CLABSI) prevention practices
- Core Strategies:
- Follow evidence-based guidance for the prevention of CLABSIs
- See Table 1 for decolonization and pathogen reduction strategies by patient type and setting including for patents with CVCs
- Core Strategies:
- Surgical site infection (SSI) prevention practices
- Core Strategies:
- Follow evidence-based guidance for the prevention of SSIs
- Core Strategies:
- Hemodialysis bloodstream infection prevention practices
- Core Strategy:
- Follow evidence-based guidance for the prevention of hemodialysis bloodstream infections
- Available guidance:
- Follow evidence-based guidance for the prevention of hemodialysis bloodstream infections
- Core Strategy:
- Ventilator-associated Pneumonia (VAP) prevention practices
- Core Strategy:
- Follow evidence-based guidance for the prevention of VAP
- Core Strategy:
2. IMPLEMENT DECOLONIZATION AND PATHOGEN REDUCTION STRATEGIES FOR HIGH RISK PATIENTS DURING HIGH RISK PERIODS
- Core Strategy:
- Pursue a strategy to reduce carriage of S. aureus among all patients admitted to intensive care units (ICUs) (see table for summary of decolonization and pathogen reduction strategies) including:
- Apply intranasal mupirocin twice a day to each naris for 5 days in conjunction with daily chlorhexidine bathing for duration of ICU admission
- Based on the results of a single trial, intranasal mupirocin may be preferred; however, intranasal iodophor could be considered as an alternative to intranasal mupirocin
- For more information see: Universal ICU Decolonization: An Enhanced Protocol. Agency for Healthcare Research and Quality (AHRQ).
- Apply intranasal mupirocin twice a day to each naris for 5 days in conjunction with daily chlorhexidine bathing for duration of ICU admission
- Pursue a strategy to reduce carriage of S. aureus among all patients admitted to intensive care units (ICUs) (see table for summary of decolonization and pathogen reduction strategies) including:
- Supplemental Strategy
- Pursue a strategy to reduce carriage of S. aureus for patients hospitalized with CVCs or midline catheters outside the ICU
- Apply intranasal mupirocin twice a day to each naris for 5 days in conjunction with daily chlorhexidine bathing while CVC or midline catheter is present
- Based on the results of a single trial intranasal mupirocin may be preferred however, intranasal iodophor could be considered as an alternative to intranasal mupirocin
- Apply intranasal mupirocin twice a day to each naris for 5 days in conjunction with daily chlorhexidine bathing while CVC or midline catheter is present
- Pursue a strategy to reduce carriage of S. aureus for patients hospitalized with CVCs or midline catheters outside the ICU
- Core Strategy:
- For patients undergoing high risk surgeries (e.g. cardiothoracic (CT), orthopedic, and neurosurgery), use an intranasal antistaphylococcal antibiotic/antiseptic (e.g. mupirocin or iodophor) and chlorhexidine wash or wipes prior to surgery.*
- Possible Regimens
- Intranasal antistaphylococcal antibiotic/antiseptic
- Mupirocin twice daily to each nare for the 5 days prior to day of surgery
OR - 2 applications of nasal Iodophor (at least 5%) to each nare within 2 hours prior to surgery
- Mupirocin twice daily to each nare for the 5 days prior to day of surgery
- Chlorhexidine
- Daily chlorhexidine wash or wipes for up to 5 days prior to surgery
- Intranasal antistaphylococcal antibiotic/antiseptic
- Possible Regimens
- For patients undergoing high risk surgeries (e.g. cardiothoracic (CT), orthopedic, and neurosurgery), use an intranasal antistaphylococcal antibiotic/antiseptic (e.g. mupirocin or iodophor) and chlorhexidine wash or wipes prior to surgery.*
- Supplement Strategy
- Consider chlorhexidine bathing or wipes for up to 5 days prior to surgery for all surgical patients*, not just those undergoing high risk surgeries
3. IMPLEMENT INTERVENTIONS TO PREVENT TRANSMISSION OF METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS (MRSA) IN ACUTE CARE
- Core Strategies
- The Centers for Disease Control and Prevention (CDC) continues to recommend placing patients colonized or infected with MRSA in private rooms and on Contact Precautions in inpatient acute care settings
- Use dedicated patient-care equipment (e.g. blood pressure cuffs, stethoscopes), and single use disposable items (e.g. single patient digital thermometer) whenever possible
- If common use of equipment for multiple patients is unavoidable, clean and disinfect such equipment before use on another patient
- Provide regular competency-based training on use of PPE and monitor adherence
- Place patients with excessive wound drainage (i.e. suggests an increased potential for extensive environmental contamination and risk of transmission) on Contact Precautions and in a private room regardless of Multi-drug resistant organisms (MDRO) carriage status
- Supplemental Strategy
- Consider active surveillance testing (screening) for MRSA on admission to acute care facilities. Screening could be limited to high risk patients (e.g., prior healthcare exposure) or admission to high risk settings (e.g., intensive care unit)
- Those found to be colonized with MRSA should be placed in private rooms and on Contact Precautions
- Active surveillance testing could be combined with decolonization or pathogen reduction strategies as described above for high risk patients (i.e. ICU patients and those outside the ICU with CVCs or Midline Catheters)
- Consider active surveillance testing (screening) for MRSA on admission to acute care facilities. Screening could be limited to high risk patients (e.g., prior healthcare exposure) or admission to high risk settings (e.g., intensive care unit)
4. DEVELOP INFRASTRUCTURE TO SUPPORT HO SA BSI PREVENTION
- Core Strategies
- Incorporate reduction of HO SA BSIs into the facility healthcare-associated infection prevention program
- Develop a multidisciplinary workgroup, including nursing, environmental services, and infection prevention to identify and implement strategies and to follow results of interventions
- Monitor facility HO SA BSI counts, and target units with highest number of HO SA BSIs for evaluation and intervention
- Provide HO SA BSI rates to senior leadership, clinical staff, and other stakeholders
- Notify appropriate individuals and facility departments about changes in the incidence (or frequency), complications (including recurrences), or severity of HO SA BSIs
- Review individual HO SA BSI episodes to assess modifiable risk factors including clinical management decisions and the use of infection control measures to identify gaps
- Educate and train all healthcare personnel on prevention practices for HO SA BSI and core infection control practices such as hand hygiene, PPE use, Standard Precautions, Contact Precautions, and environmental cleaning and disinfection
- Routinely audit and conduct competency-based assessments for core infection control practices
- Adherence to hand hygiene, Standard Precautions, and Contact Precautions
- Adequacy of room cleaning and environmental services
- Incorporate reduction of HO SA BSIs into the facility healthcare-associated infection prevention program
Table 1: Summary of Decolonization and Pathogen Reduction Strategies by Central Venous Catheter (CVC) or Midline Catheter Presence and Unit Type:
Patient Type Intensive Care Unit Non-Intensive Care Unit
CVC or Midline Catheter Present Topical chlorhexidine gluconate (at least 2%) + Intranasal antistaphylococcal antibiotic/antiseptic (e.g. mupirocin or iodophor) (core strategy) Topical chlorhexidine gluconate (at least 2%) + Intranasal antistaphylococcal antibiotic/antiseptic (e.g. mupirocin or iodophor) ( supplemental strategy)
No CVC or Midline Catheter Topical chlorhexidine gluconate (at least 2%) + Intranasal antistaphylococcal antibiotic/antiseptic (i.e. mupirocin or iodophor) (core strategy)
None (note that decolonization or pathogen reduction strategies may apply to pre-operative surgical patients outside the intensive care unit- see section 2) (CDC)
Precautions: Contact Precautions"CDC recommends the use of Contact Precautions in inpatient acute care settings for patients known to be colonized or infected with epidemiologically important multidrug-resistant organisms (MDROs) including MRSA.
From 2005 to 2014, the overall estimated incidence of invasive MRSA infections from normally sterile sites (i.e., blood, pleural fluid, etc.) in the United States declined by 40% and the estimated incidence of invasive hospital-onset MRSA infections declined by 65%. Interventions designed to decrease risk of device and procedure-associated infections and interventions to reduce transmission, like Contact Precautions and hand hygiene, both likely contributed to these decreases. The relative contribution of Contact Precautions in comparison to other interventions is unknown.
In the past 5 years, more than a dozen studies, review articles, and editorials have sought to address whether Contact Precautions should continue to be recommended for endemic MDROs like MRSA. The impact of discontinuing Contact Precautions for MRSA-colonized or infected patients has been assessed primarily in single-center studies using lower quality quasi-experimental designs that have not identified changes in MRSA infection or acquisition rates. These studies likely underestimate the impact of discontinuing Contact Precautions, including the effect on downstream adverse events (e.g., post-discharge infections).
Based on the current evidence, CDC continues to recommend the use of Contact Precautions for MRSA-colonized or infected patients. CDC will continue to evaluate the evidence on Contact Precautions as it becomes available. In addition, CDC continues to work with partners to identify and evaluate other measures to decrease transmission of MDROs in healthcare settings." (CDC)
"MRSA remains an important healthcare pathogen and the prevention of MRSA infections is a priority for CDC. CDC estimates that MRSA is responsible for more than 70,000 severe infections and 9,000 deaths per year.
Make preventing staph infections (MRSA and MSSA) a priority in healthcare facilities:
- assess your facility’s data,
- implement prevention actions, and
- evaluate progress.
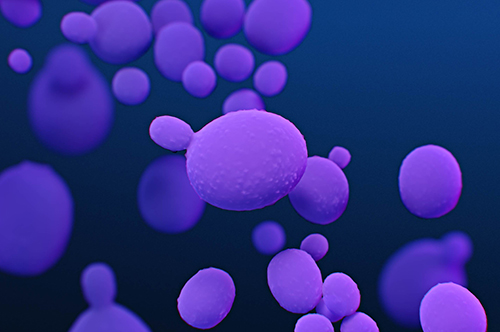
Candida auris:
"Candida auris (C. auris) is a fungus, specifically a type of yeast that can cause severe illness. It is transmitted easily among patients in healthcare facilities. It is typically resistant to antifungal treatments, which means that the medications that are designed to kill the fungus and stop infections do not work and are ineffective. Resistance to antifungals is on the rise, particularly amongst this organism." (CDC)
The following information is directly from the CDC website:
"Symptoms:
C. auris can cause infections in different parts of the body, including the bloodstream, open wounds, and ears. The symptoms depend on the location and severity of C. auris infection. Symptoms may be similar to symptoms of an infection caused by bacteria. There is not a common set of symptoms specific for C. auris infections.
Colonization:
People can carry C. auris on their skin and other body sites without having any symptoms or having an active infection. You may hear healthcare providers refer to this as ‘colonization.’ Someone who is colonized can still, as a vector (living carrier), transmit C. auris onto surfaces or objects (fomites, or nonliving carriers) that they contact, which can then spread it to other patients. This is a form of direct or indirect contact.
Diagnoses (Testing and Screening):
There are two ways that a healthcare provider can see if a patient is infected or colonized with C. auris.
Not a Threat to Healthy People:
In general, C. auris does not pose a threat to healthy individuals. The CDC typically does not recommend screening or testing family members. Family members should use alcohol-based hand sanitizer or wash their hands before entering and leaving a patient’s room and before and after contact with the patient or a patient’s medical devices.
If a family member or someone else with frequent contact with a patient with C. auris needs to receive care at a healthcare facility, they should tell the healthcare provider.
Patients and family members should talk to a healthcare provider or the facility’s management if they are concerned about infection protection and control.
Risk Factors:
C. auris mostly affects patients with severe underlying medical conditions and requiring complex medical care. Patients with invasive medical devices like breathing tubes, feeding tubes, catheters in a vein, or urinary catheters tend to be at increased risk for getting C. auris and developing an infection.
Healthy people without these risk factors, including healthcare workers and family members, have a low risk for getting infected with C. auris.
Resistance and Treatment:
C. auris is often resistant to commonly used antifungal medications, most C. auris infections are treatable with a class of antifungal medicines called echinocandins.
However, some C. auris strains have been resistant to all three main classes of antifungal medicines, meaning none are able to treat the infection. In this situation, multiple antifungal medicines or newer antifungals may be used to treat the infection.
Patients who are colonized (have C. auris detected on their body but do not have symptoms of infection) should not be treated with antifungals for C. auris. There is no evidence that this prevents sickness.
Outcomes:
C. auris can cause a variety of infections from superficial (skin) infections to more severe, life-threatening infections. C. auris is the most dangerous when it causes invasive infections, such as bloodstream infections. However, many of the patients who become sick with C. auris are usually already very sick, which makes it hard to know how much C. auris contributed to their death.
Preventing Spread:
Patient care goes beyond treating or managing an illness or condition. Sick patients face added risks of getting new infections in healthcare settings, even when staff follow prevention guidelines.
If a patient is colonized or infected, healthcare providers take special steps to prevent the spread of C. auris, including placing the patient in a room separated from those at risk, cleaning the rooms with special disinfectant products, and wearing gloves and gowns to deliver care.
Healthcare facilities should be informed if an incoming patient has ever:
Why is Candida auris a problem?
Stopping the spread of Candida auris:
"The CDC is working with public health partners, healthcare personnel, and laboratories to stop the spread of C. auris in healthcare settings. Here’s how CDC is asking everyone to help:
CDC and public health partners are working hard to better understand C. auris and answer the following questions so that we can continue to help protect people from this serious infection:
The following information is directly from the CDC website:
"Symptoms:
C. auris can cause infections in different parts of the body, including the bloodstream, open wounds, and ears. The symptoms depend on the location and severity of C. auris infection. Symptoms may be similar to symptoms of an infection caused by bacteria. There is not a common set of symptoms specific for C. auris infections.
Colonization:
People can carry C. auris on their skin and other body sites without having any symptoms or having an active infection. You may hear healthcare providers refer to this as ‘colonization.’ Someone who is colonized can still, as a vector (living carrier), transmit C. auris onto surfaces or objects (fomites, or nonliving carriers) that they contact, which can then spread it to other patients. This is a form of direct or indirect contact.
Diagnoses (Testing and Screening):
There are two ways that a healthcare provider can see if a patient is infected or colonized with C. auris.
- Colonization screening– a healthcare provider swabs the patient’s skin by rubbing a swab near the armpits and groin and sends the swab to a laboratory for testing, similar to how this is done for MRSA screening.
- Clinical specimen testing– If a patient is showing symptoms of an infection of unknown cause, a healthcare provider may collect a clinical sample, like blood or urine. They usually test for many types of infections including those caused by bacteria and the results may show that the patient has C. auris. Cultures are grown on fungal media, then tested for identification. The Vitek 2 yeast card and the mass spectrophotomter are ways that this organism can be identified.
Not a Threat to Healthy People:
In general, C. auris does not pose a threat to healthy individuals. The CDC typically does not recommend screening or testing family members. Family members should use alcohol-based hand sanitizer or wash their hands before entering and leaving a patient’s room and before and after contact with the patient or a patient’s medical devices.
If a family member or someone else with frequent contact with a patient with C. auris needs to receive care at a healthcare facility, they should tell the healthcare provider.
Patients and family members should talk to a healthcare provider or the facility’s management if they are concerned about infection protection and control.
Risk Factors:
C. auris mostly affects patients with severe underlying medical conditions and requiring complex medical care. Patients with invasive medical devices like breathing tubes, feeding tubes, catheters in a vein, or urinary catheters tend to be at increased risk for getting C. auris and developing an infection.
Healthy people without these risk factors, including healthcare workers and family members, have a low risk for getting infected with C. auris.
Resistance and Treatment:
C. auris is often resistant to commonly used antifungal medications, most C. auris infections are treatable with a class of antifungal medicines called echinocandins.
However, some C. auris strains have been resistant to all three main classes of antifungal medicines, meaning none are able to treat the infection. In this situation, multiple antifungal medicines or newer antifungals may be used to treat the infection.
Patients who are colonized (have C. auris detected on their body but do not have symptoms of infection) should not be treated with antifungals for C. auris. There is no evidence that this prevents sickness.
Outcomes:
C. auris can cause a variety of infections from superficial (skin) infections to more severe, life-threatening infections. C. auris is the most dangerous when it causes invasive infections, such as bloodstream infections. However, many of the patients who become sick with C. auris are usually already very sick, which makes it hard to know how much C. auris contributed to their death.
Preventing Spread:
Patient care goes beyond treating or managing an illness or condition. Sick patients face added risks of getting new infections in healthcare settings, even when staff follow prevention guidelines.
If a patient is colonized or infected, healthcare providers take special steps to prevent the spread of C. auris, including placing the patient in a room separated from those at risk, cleaning the rooms with special disinfectant products, and wearing gloves and gowns to deliver care.
Healthcare facilities should be informed if an incoming patient has ever:
- Tested positive for C. auris, with or without symptoms.
- Was exposed to another patient with C. auris.
- Was in a facility where an outbreak was occurring.
Why is Candida auris a problem?
- "It causes serious infections. C. auris can cause bloodstream infections (sepsis or bacteremia) and even death, particularly in hospital and nursing home patients with serious medical problems. More than 1 in 3 patients with invasive C. auris infection (for example, an infection that affects the blood, heart, or brain) die.
- It’s often resistant to medicines. Antifungal medicines commonly used to treat Candida infections often don’t work for Candida auris. Some C. auris infections have been resistant to all three types of antifungal medicines.
- It’s becoming more common. Although C. auris was just discovered in 2009, it has spread quickly and caused infections in more than a dozen countries.
- It’s difficult to identify. C. auris can be misidentified as other types of fungi unless specialized laboratory technology is used. This misidentification might lead to a patient getting the wrong treatment.
- It can spread in hospitals and nursing homes. C. auris has caused outbreaks in healthcare facilities and can spread through contact with affected patients and contaminated surfaces or equipment. Good hand hygiene and cleaning in healthcare facilities is important because C. auris can live on surfaces for several weeks." (CDC)
Stopping the spread of Candida auris:
"The CDC is working with public health partners, healthcare personnel, and laboratories to stop the spread of C. auris in healthcare settings. Here’s how CDC is asking everyone to help:
- Family members and other close contacts of patients with C. auris
- Clean your hands with hand sanitizer or soap and water before and after touching a patient with C. auris or equipment in his or her room.
- Remind healthcare personnel to clean their hands.
- Laboratory staff, healthcare personnel, and public health officials
- Know when to suspect C. auris and how to properly identify it.
- Report cases quickly to public health departments.
- For healthcare personnel, clean hands correctly and use precautions like wearing gowns and gloves to prevent spread.
- Clean patient rooms thoroughly with a disinfectant that works against C. auris.
- Investigate C. auris cases quickly and determine additional ways to prevent spread.
- See Recommendations for Identification, Treatment, and Infection Prevention and control of Candida auris.
CDC and public health partners are working hard to better understand C. auris and answer the following questions so that we can continue to help protect people from this serious infection:
- Why is C. auris resistant to antifungal medicines? This is due to genes, but scientists are still researching which ones.
- Why did C. auris start causing infections in recent years?
- Where did C. auris originally come from, and why has it appeared in many regions of the world at the same time?
- Advising healthcare personnel and infection control staff on ways to stop the spread of C. auris and continually updating this guidance as we learn more about the infection.
- Working with state and local health agencies, healthcare facilities, and clinical microbiology laboratories to ensure that laboratories are using proper methods to detect C. auris.
- Testing C. auris strains to monitor for resistance to antifungal medicines.
- Examining the DNA of C. auris strains using whole genome sequencing to better understand how this germ is spreading in the United States and around the world.
- Working with public health partners in the United States and internationally to learn more about how C. auris spreads in healthcare facilities and to eliminate it from those facilities." (CDC)
| candida_auris.pptx | |
| File Size: | 5519 kb |
| File Type: | pptx |
Central line-Associated Bloodstream Infections:
According to the CDC, a central venous catheter, is also also called as a central line. It is a tube that doctors or nurses place in a large vein in the neck, chest, groin, or arm. It is put into place in order to administer fluids, blood, or medications or to do medical tests that need to be drawn quickly. Some of these medications treat heart conditions, cancer, infections, or treat pain. Some of the fluids are nutritional.
These tubes are long, flexible catheters that drain in or near the heart, in order for the catheter to give the required treatment within seconds. You may be more familiar with standard intravenous lines (IVs). Central lines are very different from standard IVs that are used to give medicine into a vein near the skin’s surface, usually for short periods of time. A central venous catheter can remain for weeks or months, and some patients receive treatment through the line several times a day. Central venous catheters are important in treating many conditions, particularly in intensive care units (ICUs) (CDC).
There are a few types of central venous catheters. Healthcare providers choose the type that is best for each patient’s individual case.
A central line-associated bloodstream infection (CLABSI) is a serious infection! It occurs when germs (usually bacteria or viruses) enter the bloodstream through the central line, resulting in bacteremia or sepsis. Healthcare providers must follow a strict protocol when inserting the line to make sure the line remains sterile and a CLABSI does not occur. In addition to inserting the central line properly, healthcare providers must use strict infection control practices every time they check the line or change the dressing. Patients who get a CLABSI have a fever, and might also have red skin and soreness around the central line, their white blood cell count might be elevated, and their blood pressure might be low (hypotension), which is serious, because it can cause septic shock. If this happens, healthcare providers can do tests to learn if there is an infection present by ordering microbiology and hematology (blood) tests to look for the presence of microbes and to see if there is an elevation in the white blood cell count.
Prevention is key! According to the CDC guidelines, here are some things that providers can do to help prevent these types of infections:
Healthcare providers can take the following steps to help prevent CLABSIs:
These tubes are long, flexible catheters that drain in or near the heart, in order for the catheter to give the required treatment within seconds. You may be more familiar with standard intravenous lines (IVs). Central lines are very different from standard IVs that are used to give medicine into a vein near the skin’s surface, usually for short periods of time. A central venous catheter can remain for weeks or months, and some patients receive treatment through the line several times a day. Central venous catheters are important in treating many conditions, particularly in intensive care units (ICUs) (CDC).
There are a few types of central venous catheters. Healthcare providers choose the type that is best for each patient’s individual case.
- "A peripherally inserted central catheter (PICC) line is placed into a vein in the arm.
- A tunneled catheter is surgically placed into a vein in the chest or neck and then passed under the skin. One end of the catheter comes out through the skin so medicines can be given right into the catheter. This is beneficial for patients needing chemotherapy or other cancer medications, for example. It stays in for a long period of time.
- An implanted port is similar to a tunneled catheter, but an implanted port is placed entirely under the skin. Medicines are given by a needle placed through the skin into the catheter. An implanted port is not as visible as a tunneled catheter, does not require as much daily care, and does not get in the way of a patient’s regular activities as much as a PICC line or a tunneled catheter might." (CDC)
A central line-associated bloodstream infection (CLABSI) is a serious infection! It occurs when germs (usually bacteria or viruses) enter the bloodstream through the central line, resulting in bacteremia or sepsis. Healthcare providers must follow a strict protocol when inserting the line to make sure the line remains sterile and a CLABSI does not occur. In addition to inserting the central line properly, healthcare providers must use strict infection control practices every time they check the line or change the dressing. Patients who get a CLABSI have a fever, and might also have red skin and soreness around the central line, their white blood cell count might be elevated, and their blood pressure might be low (hypotension), which is serious, because it can cause septic shock. If this happens, healthcare providers can do tests to learn if there is an infection present by ordering microbiology and hematology (blood) tests to look for the presence of microbes and to see if there is an elevation in the white blood cell count.
Prevention is key! According to the CDC guidelines, here are some things that providers can do to help prevent these types of infections:
Healthcare providers can take the following steps to help prevent CLABSIs:
- "Follow recommended central line insertion practices to prevent infection when the central line is placed, including:
- Perform hand hygiene.
- Apply appropriate skin antiseptic.
- Ensure that the skin prep agent has completely dried before inserting the central line.
- Use all five maximal sterile barrier precautions:
- Sterile gloves
- Sterile gown
- Cap
- Mask
- Large sterile drape
- Perform hand hygiene.
- Once the central line is in place:
- Follow recommended central line maintenance practices.
- Wash their hands with soap and water for a minimum of 20 seconds, lathering up, rinsing, or apply an alcohol-based hand rub (ABHR) before and after touching the line.
- Follow recommended central line maintenance practices.
- Remove a central line as soon as it is no longer needed. The sooner a catheter is removed, the less likely the chance of infection." Keep this in mind while planning the patient's discharge summaries.
- "Research the hospital, if possible, to learn about its CLABSI rate.
- Speak up about any concerns so that healthcare personnel are reminded to follow the best infection prevention practices.
- Ask a healthcare provider if the central line is absolutely necessary. If so, ask them to help you understand the need for it and how long it will be in place.
- Pay attention to the bandage and the area around it. If the bandage comes off or if the bandage or area around it is wet or dirty, tell a healthcare worker right away.
- Don’t get the central line or the central line insertion site wet.
- Tell a healthcare worker if the area around the catheter is sore or red or if the patient has a fever or chills.
- Do not let any visitors touch the catheter or tubing.
- The patient should avoid touching the tubing as much as possible.
- In addition, everyone visiting the patient must wash their hands—before and after they visit."