MALDI-TOF Mass spectrometry:
MALDI-TOF Mass spectrometry is one of the newest analytical technologies utilized in Microbiology as a rapid means of identifying various microbes. Scientifically based on Physics and Biochemistry, this analytical instrument measures the mass-to-charge ratio of ions. This is presented as a mass spectrum, hence the name. It presents a plot intensity of the ion signal and determines isotopes and the mass of particles and molecules.
Samples Being Tested:
Samples must be tested from pure cultures (remember pure cultures from Microbiology Introduction Tab). This means that this may take a couple of days or more before it is ready to be tested on the MALDI-TOF Mass Spec. Samples are cultured, then the microbe of choice is subcultured onto a another more specific plate to isolate a pure culture. From the pure culture, a "pick-me" pen or small biological loop is used to swipe a small portion of the colony and smear it in a circle on the stainless steel target slide. If it is bacteria, 1.00 uL's of matrix is added into the circle over the sample and allowed to completely air dry (it dries quickly). If it is yeast being tested, .50 uL's of formic acid is added first, allowed to dry (takes longer), then 1.00 uL's of matrix is added on top of that. When the slide is dry, it is ready to go onto the MALDI-TOF Mass Spectrophotometer. Each slide has a unique barcode that is scanned into the computer LIS system. Each slide has 3 acquisitions. Each acquisition has 16 circles, or space for 8 samples (each sample is performed in duplicate to increase chances of rapid ID), with a smaller circle in the middle for the calibrator. E. coli is used as the calibrator and if it fails, the entire acquisition will fail and samples need to be repeated. The same matrix of 1.00 uL's is added to the calibrator circle in the middle. One to three acquisitions at a time can be performed. Each patient sample also has a barcode and that is scanned in and each sample is assigned a place on the slide. It takes anywhere from about 30 minutes to 1.5 hours for a slide to be read and results to appear. Quality control (QC) is performed once per day and patient results are not released unless the QC passes and is good. One bacteria sample and one yeast sample is used. If AFB and Nocardia are being tested, then QC is performed at that time with a sample organism. It is important to note that on occasion, if the Mass Spec. is having difficulty, it may be down for awhile or undergoing periodic maintenance, and results will take longer. Typically there is a backup method, such as identification cards, or samples must be sent out to a reference lab.
Computer Software and Database:
Middleware software records the information and results. Results are compared with an internal database with thousands of validated identifications. If the ID is new to your lab, it has to be sent out for verification, but results will still show up in the computer for the patient. Results are then analyzed for accuracy, and accepted (sent across to the laboratory LIS) or rejected (discarded) or repeated. The bench tech. is the one who ultimately enables or disables the result from showing up or manually enters the result and supporting information so that the healthcare workers, such as nurses, doctors, PA's, and nurse practitioners and others may see it and access it. For questions about results, the person working on that bench or culture is the one to request and can help you. This is one of the main technologies healthcare workers will be relying on the function properly for the rapid and accurate ID of microbes that may be causing disease or symptoms in the patient, to aid in diagnosis and treatment. Following this, some samples are set up on an analyzer such as Vitek 2 for susceptibility (antibiotic) testing, and then those results will cross over into the LIS, revealing which antibiotics the bug is sensitive to and which ones it is resistant to. You can request specific drugs for testing or it can be sent out for further testing.
Mass Spec. Technology: How the Testing Works:
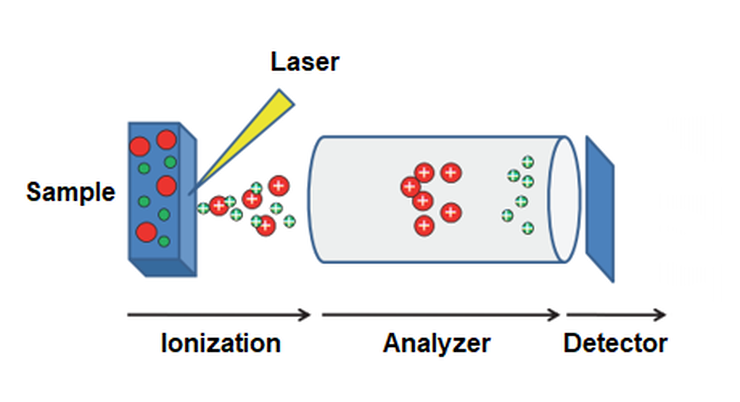
On the Mass Spec, samples tested may be solids, liquids, or gas hit with electrons to become ionized (electrically charged molecules) (10). There is an electron multiplier that results in ionization. Inside the instrument there are 3 key parts:
The mass analyzer (TOF) contains both electric and magnetic fields, which place a force on the ions, causing them to become charged and increase or decrease in speed or change direction, based on Newton's Second Law of Motion (F=ma). There is a stage of acceleration and the lighter charged particles reach the reflectron and detectors faster than the heavier ones.
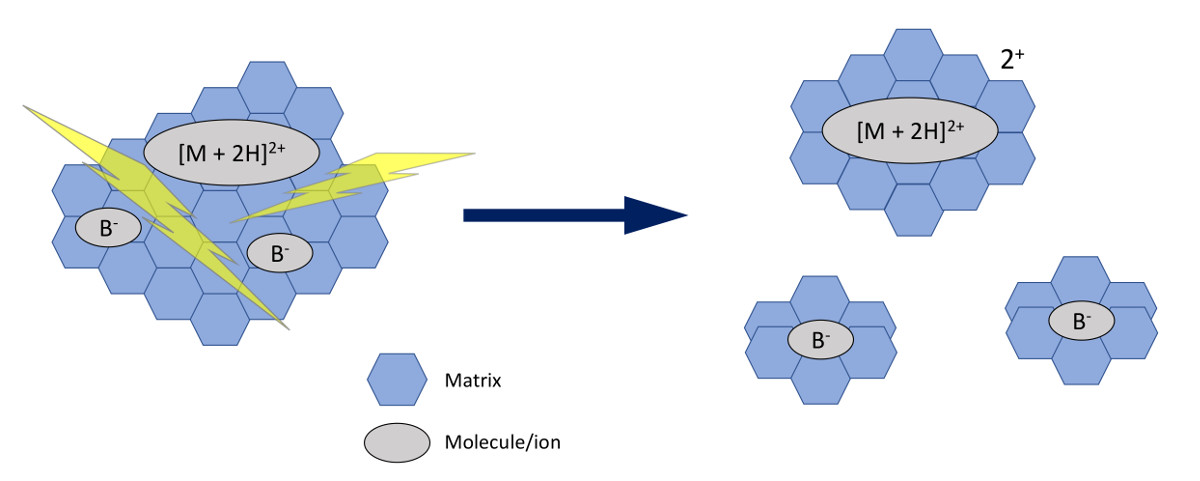
Mass spectrometry was invented in 1985. Sample is mixed with matrix material (usually an ultrafine crystallized metal and a liquid) and applied to a metal plate (stainless steel). The first matrix consisted of ioinized amino acids alanine and tryptophan, which served as an absorber. Currently, matrix consists of one of three highly purified acids: 3,5 dimethoxy-4-hydroxycinnamic acid (sinapinic acid), a-cyano-4-hydroxycinnamic acid (CHCA), or 2,5 dihydroxybenzoic acid dissolved to form an acidic solution with highly purified water plus an organic solvent (ethanol or acetonitrile) plus a counter ion source (trifluoroacetic acid) to generate M + H ions, plus a chromophore. This solution is mixed with the sample, allowing it to contain BOTH hydrophilic (water-loving) AND hydrophobic (water-resisting) portions, resulting in co-crystallization, which is key to the success of this technology.
The Matrix:
Sinapinic acid is naturally occurring hydroxycinnamic acid that is derived from sinapine, an alkaline amine found naturally in black mustard seeds (6). It is chosen for MALDI-TOF since it can readily absorb laser radiation and donate protons (H+) to the analyte being identified by it (4)(5). Swedish botanist and scientist Carl Linnaeus and German botanist and scientist Karl Koch described the black mustard seed in 1833 (2)(3). It was first used in folk medicine in the UK to treat individuals with colds by giving them a hot mustard bath (1). It was also mixed with honey and used as a cough suppressant throughout Europe and in Canada to treat respiratory infections (1). It was used to treat muscle pain (1). Prior to modern medicine, the seeds were ground up and mixed with water and flour to create a "hot" paste that was applied to the skin over muscles in pain, or to place on the chest and back of an individual suffering from a cough or respiratory symptoms (1). It's currently used as a spice, mostly in India, and is often part of curry dishes. It adds "heat" to spicy dishes, is added to fermenting grapes and vinegars during the fermentation process to give them "heat" or "edge", and is part of the UK's "hot" mustard. Believe it or not, tasting a little bit of mustard can also aid in relieving nausea, particularly due to pregnancy. It's amazing how far we've come with biochemicals and medicine, especially with this compound, which is now a critical component of the MALDI-TOF process (matrix), which will not work without it, and aids in the rapid identification of bacteria, yeasts, mycobacteria, and some molds.
CHCA is derived from cinnamic acid, which is an organic compound and an aromatic compound with a 6-carbon skeleton. It has the following formula: C6H5CH=CHCOOH. Naturally, it is a white crystalline powder that is pretty soluble in water and organic solvents. It is found in many plants and in cinnamon (oil of cinnamon), balsam, aloe, and shea butter (9). In MALDI-TOF Mass Spec., it is used as a matrix for peptides (from proteins), lipids, and nucleotides (building blocks of DNA and RNA) (7)(8).
The Laser(s):
There is a UV pulsed laser inside which irradiates the sample, triggering ablation and desorption of sample/matrix. Originally, this laser consisted of cobalt in glycerol and nitrogen and pulsed at 266, 337 or 355 nm. Currently, most are UV lasers consisting of nitrogen (N) pulsed at 337 nm and frequency-tripled and quadrupled Nd: YAG lasers that pulse at 355 nm and 266 nm, a Mid-IR Optical Parametric Oscillator, and 10.6 micrometers of CO2. The matrices are both polar and nonpolar in nature.
Ionization:
Analyte molecules are ionized in a hot plume of ablated gases, resulting in protonation and deprotonation (H+ ions). Ions are accelerated into the mass spectrometer. The advantages of mass spectrometry are that it is rapid, reliable, low-cost, it detects proteins, it detects peptides, it detects lipids, it detects saccharides, it detects other macromolecules, and it can detect multi-drug-resistant organisms (MDRO's) in as little as 3 hours, including carbapenemases and B-lactams, resulting in more rapid treatment for the patient. This technology produces amazingly accurate and precise results and rapid turnaround time.
Terminology:
Samples Being Tested:
Samples must be tested from pure cultures (remember pure cultures from Microbiology Introduction Tab). This means that this may take a couple of days or more before it is ready to be tested on the MALDI-TOF Mass Spec. Samples are cultured, then the microbe of choice is subcultured onto a another more specific plate to isolate a pure culture. From the pure culture, a "pick-me" pen or small biological loop is used to swipe a small portion of the colony and smear it in a circle on the stainless steel target slide. If it is bacteria, 1.00 uL's of matrix is added into the circle over the sample and allowed to completely air dry (it dries quickly). If it is yeast being tested, .50 uL's of formic acid is added first, allowed to dry (takes longer), then 1.00 uL's of matrix is added on top of that. When the slide is dry, it is ready to go onto the MALDI-TOF Mass Spectrophotometer. Each slide has a unique barcode that is scanned into the computer LIS system. Each slide has 3 acquisitions. Each acquisition has 16 circles, or space for 8 samples (each sample is performed in duplicate to increase chances of rapid ID), with a smaller circle in the middle for the calibrator. E. coli is used as the calibrator and if it fails, the entire acquisition will fail and samples need to be repeated. The same matrix of 1.00 uL's is added to the calibrator circle in the middle. One to three acquisitions at a time can be performed. Each patient sample also has a barcode and that is scanned in and each sample is assigned a place on the slide. It takes anywhere from about 30 minutes to 1.5 hours for a slide to be read and results to appear. Quality control (QC) is performed once per day and patient results are not released unless the QC passes and is good. One bacteria sample and one yeast sample is used. If AFB and Nocardia are being tested, then QC is performed at that time with a sample organism. It is important to note that on occasion, if the Mass Spec. is having difficulty, it may be down for awhile or undergoing periodic maintenance, and results will take longer. Typically there is a backup method, such as identification cards, or samples must be sent out to a reference lab.
Computer Software and Database:
Middleware software records the information and results. Results are compared with an internal database with thousands of validated identifications. If the ID is new to your lab, it has to be sent out for verification, but results will still show up in the computer for the patient. Results are then analyzed for accuracy, and accepted (sent across to the laboratory LIS) or rejected (discarded) or repeated. The bench tech. is the one who ultimately enables or disables the result from showing up or manually enters the result and supporting information so that the healthcare workers, such as nurses, doctors, PA's, and nurse practitioners and others may see it and access it. For questions about results, the person working on that bench or culture is the one to request and can help you. This is one of the main technologies healthcare workers will be relying on the function properly for the rapid and accurate ID of microbes that may be causing disease or symptoms in the patient, to aid in diagnosis and treatment. Following this, some samples are set up on an analyzer such as Vitek 2 for susceptibility (antibiotic) testing, and then those results will cross over into the LIS, revealing which antibiotics the bug is sensitive to and which ones it is resistant to. You can request specific drugs for testing or it can be sent out for further testing.
Mass Spec. Technology: How the Testing Works:
On the Mass Spec, samples tested may be solids, liquids, or gas hit with electrons to become ionized (electrically charged molecules) (10). There is an electron multiplier that results in ionization. Inside the instrument there are 3 key parts:
- an ion source, which converts parts of the sample to ions (MALDI: matrix-assisted laser desorption/ionization)
- a mass analyzer through which ions are targeted through and sorted by (mass-to-charge ratio) (TOF: time-of-flight)
- a detector in which ions enter after they are sorted and are recorded (the charge or current is recorded)
- "Peaks"
The mass analyzer (TOF) contains both electric and magnetic fields, which place a force on the ions, causing them to become charged and increase or decrease in speed or change direction, based on Newton's Second Law of Motion (F=ma). There is a stage of acceleration and the lighter charged particles reach the reflectron and detectors faster than the heavier ones.
Mass spectrometry was invented in 1985. Sample is mixed with matrix material (usually an ultrafine crystallized metal and a liquid) and applied to a metal plate (stainless steel). The first matrix consisted of ioinized amino acids alanine and tryptophan, which served as an absorber. Currently, matrix consists of one of three highly purified acids: 3,5 dimethoxy-4-hydroxycinnamic acid (sinapinic acid), a-cyano-4-hydroxycinnamic acid (CHCA), or 2,5 dihydroxybenzoic acid dissolved to form an acidic solution with highly purified water plus an organic solvent (ethanol or acetonitrile) plus a counter ion source (trifluoroacetic acid) to generate M + H ions, plus a chromophore. This solution is mixed with the sample, allowing it to contain BOTH hydrophilic (water-loving) AND hydrophobic (water-resisting) portions, resulting in co-crystallization, which is key to the success of this technology.
The Matrix:
Sinapinic acid is naturally occurring hydroxycinnamic acid that is derived from sinapine, an alkaline amine found naturally in black mustard seeds (6). It is chosen for MALDI-TOF since it can readily absorb laser radiation and donate protons (H+) to the analyte being identified by it (4)(5). Swedish botanist and scientist Carl Linnaeus and German botanist and scientist Karl Koch described the black mustard seed in 1833 (2)(3). It was first used in folk medicine in the UK to treat individuals with colds by giving them a hot mustard bath (1). It was also mixed with honey and used as a cough suppressant throughout Europe and in Canada to treat respiratory infections (1). It was used to treat muscle pain (1). Prior to modern medicine, the seeds were ground up and mixed with water and flour to create a "hot" paste that was applied to the skin over muscles in pain, or to place on the chest and back of an individual suffering from a cough or respiratory symptoms (1). It's currently used as a spice, mostly in India, and is often part of curry dishes. It adds "heat" to spicy dishes, is added to fermenting grapes and vinegars during the fermentation process to give them "heat" or "edge", and is part of the UK's "hot" mustard. Believe it or not, tasting a little bit of mustard can also aid in relieving nausea, particularly due to pregnancy. It's amazing how far we've come with biochemicals and medicine, especially with this compound, which is now a critical component of the MALDI-TOF process (matrix), which will not work without it, and aids in the rapid identification of bacteria, yeasts, mycobacteria, and some molds.
CHCA is derived from cinnamic acid, which is an organic compound and an aromatic compound with a 6-carbon skeleton. It has the following formula: C6H5CH=CHCOOH. Naturally, it is a white crystalline powder that is pretty soluble in water and organic solvents. It is found in many plants and in cinnamon (oil of cinnamon), balsam, aloe, and shea butter (9). In MALDI-TOF Mass Spec., it is used as a matrix for peptides (from proteins), lipids, and nucleotides (building blocks of DNA and RNA) (7)(8).
The Laser(s):
There is a UV pulsed laser inside which irradiates the sample, triggering ablation and desorption of sample/matrix. Originally, this laser consisted of cobalt in glycerol and nitrogen and pulsed at 266, 337 or 355 nm. Currently, most are UV lasers consisting of nitrogen (N) pulsed at 337 nm and frequency-tripled and quadrupled Nd: YAG lasers that pulse at 355 nm and 266 nm, a Mid-IR Optical Parametric Oscillator, and 10.6 micrometers of CO2. The matrices are both polar and nonpolar in nature.
Ionization:
Analyte molecules are ionized in a hot plume of ablated gases, resulting in protonation and deprotonation (H+ ions). Ions are accelerated into the mass spectrometer. The advantages of mass spectrometry are that it is rapid, reliable, low-cost, it detects proteins, it detects peptides, it detects lipids, it detects saccharides, it detects other macromolecules, and it can detect multi-drug-resistant organisms (MDRO's) in as little as 3 hours, including carbapenemases and B-lactams, resulting in more rapid treatment for the patient. This technology produces amazingly accurate and precise results and rapid turnaround time.
Terminology:
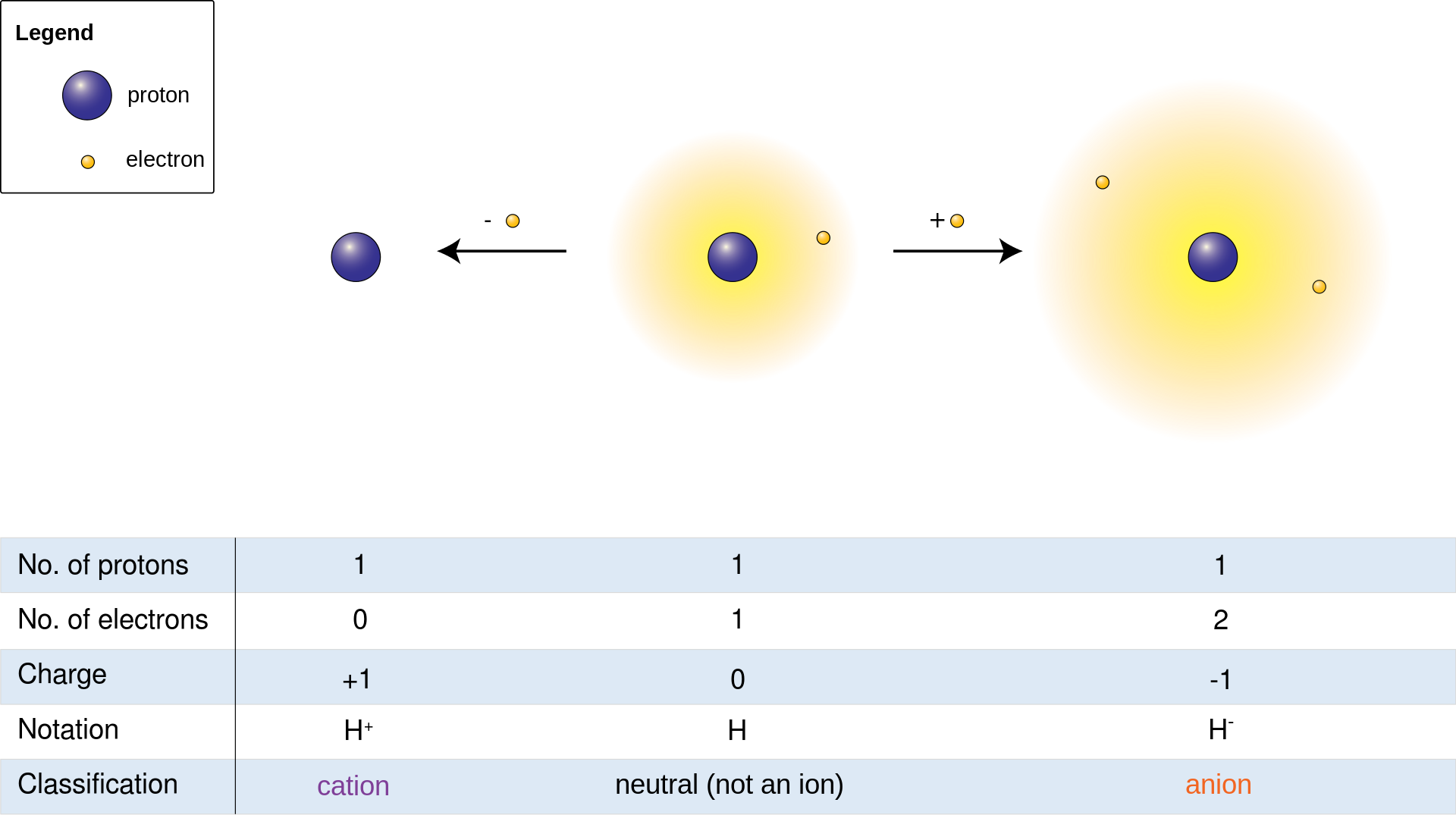
- Ion-an electrically charged atom, particle or molecule which is highly reactive in its gas-like state and will readily react with other ions of opposite charge
- Cation-a positively charged atom, particle or molecule
- Anion-a negatively charged atom, particle or molecule
- Ionization-occurs in a fluid (liquid) or gas; Ion pairs are created via spontaneous molecule collisions, each pair consisting of a free electron and a positive ion; occurs as an atom loses or gains electrons
- Chemical Ionization-the soft ionization technique used in mass spectrophotometry that was first introduced in 1966, and is a gaseous ion-molecule type of chemistry; uses reagent gas (water, ammonia, methane or isobutane) under atmospheric pressure to react with an analyte, which creates an ionization plasma
- Ion Source-a device that creates ions using high voltage and temperature within the Mass Spectrophotometer
- Proton-a positively charged particle in an atom that makes up the nucleus
- Electron-a negatively charged particle in an atom that rotates around the nucleus
- Neutron-a neutral particle in an atom (no charge) that makes up the nucleus
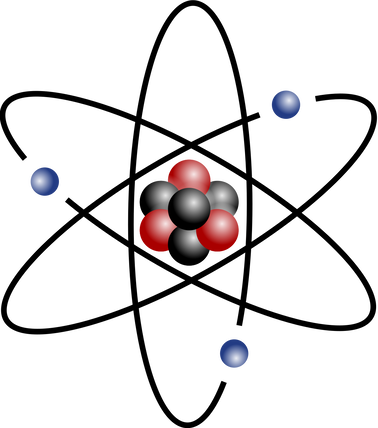
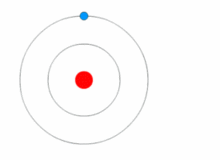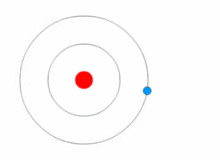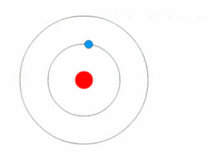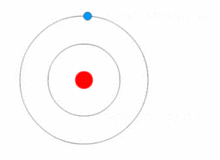
https://upload.wikimedia.org/wikipedia/commons/thumb/6/6f/Stylised_atom_with_three_Bohr_model_orbits_and_stylised_nucleus.svg/1200px-Stylised_atom_with_three_Bohr_model_orbits_and_stylised_nucleus.svg.png; An atom; Protons and neutrons make up the nucleus in the center; electrons rotate around the nucleus and are much smaller particles
references:
(1) Reader's Digest Field Guide to the Wild Flowers of Britain. Reader's Digest. 1981. p. 41. ISBN 9780276002175.
(2) "Brassica nigra (L.) K.Koch is an accepted name". theplantlist.org. 23 March 2012. Retrieved 9 November 2017.
(3) "Brassicaceae Brassica nigra (L.) W.D.J.Koch". ipni.org. Retrieved 9 November 2017.
(4) Beavis RC, Chait BT (1989). "Matrix-assisted laser-desorption mass spectrometry using 355 nm radiation". Rapid Commun. Mass Spectrom. 3 (12): 436–9. doi:10.1002/rcm.1290031208. PMID 2520224.
(5) Beavis RC, Chait BT (1989). "Cinnamic acid derivatives as matrices for ultraviolet laser desorption mass spectrometry of proteins". Rapid Commun. Mass Spectrom. 3 (12): 432–5. doi:10.1002/rcm.1290031207. PMID 2520223.
(6) Tzagoloff, A. (1963). "Metabolism of Sinapine in Mustard Plants. I. Degradation of Sinapine into Sinapic Acid & Choline". Plant Physiology. 38 (2): 202–206. doi:10.1104/pp.38.2.202. PMC 549906. PMID 16655775.
(7) Beavis, R. C.; Chaudhary, T.; Chait, B. T. (1992). "-α-Cyano-4-hydroxycinnamic acid as a matrix for matrix-assisted laser desorption mass spectrometry". Org. Mass Spectrom. 27 (2): 156–8. doi:10.1002/oms.1210270217
(8) Franz Hillenkamp; Jasna Peter-Katalinic (3 October 2013). MALDI MS: A Practical Guide to Instrumentation, Methods and Applications. Wiley. pp. 110–. ISBN 978-3-527-67373-5.
(9) Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123
(10) Hillenkamp, Franz; Karas, Michael; Beavis, Ronald C.; Chait, Brian T. (1991). "Matrix-assisted laser desorption/ionization mass spectrometry of biopolymers". Analytical Chemistry. 63 (24): 1193A–1203A. doi:10.1021/ac00024a002. ISSN 0003-2700. PMID 1789447
(2) "Brassica nigra (L.) K.Koch is an accepted name". theplantlist.org. 23 March 2012. Retrieved 9 November 2017.
(3) "Brassicaceae Brassica nigra (L.) W.D.J.Koch". ipni.org. Retrieved 9 November 2017.
(4) Beavis RC, Chait BT (1989). "Matrix-assisted laser-desorption mass spectrometry using 355 nm radiation". Rapid Commun. Mass Spectrom. 3 (12): 436–9. doi:10.1002/rcm.1290031208. PMID 2520224.
(5) Beavis RC, Chait BT (1989). "Cinnamic acid derivatives as matrices for ultraviolet laser desorption mass spectrometry of proteins". Rapid Commun. Mass Spectrom. 3 (12): 432–5. doi:10.1002/rcm.1290031207. PMID 2520223.
(6) Tzagoloff, A. (1963). "Metabolism of Sinapine in Mustard Plants. I. Degradation of Sinapine into Sinapic Acid & Choline". Plant Physiology. 38 (2): 202–206. doi:10.1104/pp.38.2.202. PMC 549906. PMID 16655775.
(7) Beavis, R. C.; Chaudhary, T.; Chait, B. T. (1992). "-α-Cyano-4-hydroxycinnamic acid as a matrix for matrix-assisted laser desorption mass spectrometry". Org. Mass Spectrom. 27 (2): 156–8. doi:10.1002/oms.1210270217
(8) Franz Hillenkamp; Jasna Peter-Katalinic (3 October 2013). MALDI MS: A Practical Guide to Instrumentation, Methods and Applications. Wiley. pp. 110–. ISBN 978-3-527-67373-5.
(9) Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123
(10) Hillenkamp, Franz; Karas, Michael; Beavis, Ronald C.; Chait, Brian T. (1991). "Matrix-assisted laser desorption/ionization mass spectrometry of biopolymers". Analytical Chemistry. 63 (24): 1193A–1203A. doi:10.1021/ac00024a002. ISSN 0003-2700. PMID 1789447