antimicrobial susceptibility testing: kirby-bauer (kb) and e-test
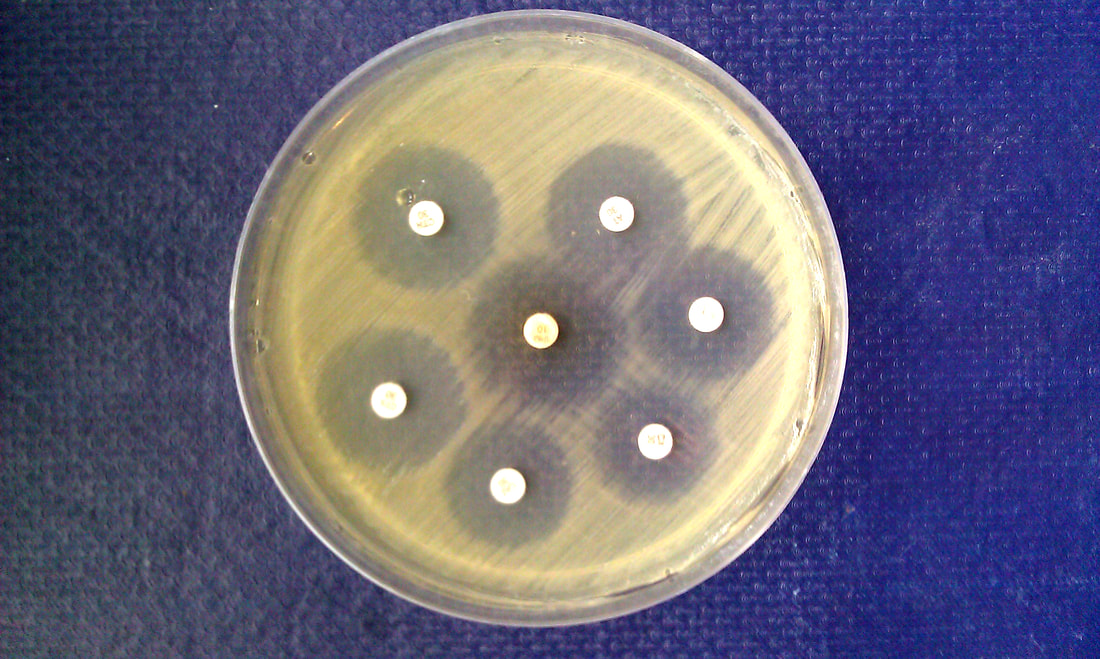
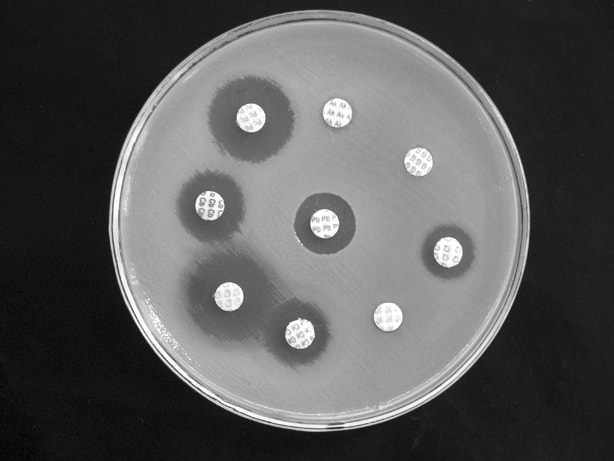
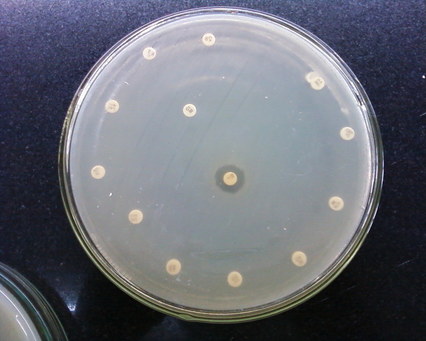
Antimicrobial susceptibility testing is a standardized method. It measures the effectiveness of antibiotics on bacteria and aids in proper physician administration of the correct antibiotic. The Kirby-Bauer test, or disk diffusion test, is a standard tool to measure the effectiveness of antibiotics against pathogens. Antimicrobial-impregnated disks, as well as KB agar plates, are refrigerated and taken out just prior to inoculation (15 minutes) and brought to room temperature. A McFarland standard solution is prepared and swabbed across the surface of the plate to create a bacterial lawn of growth. Too much or too little can produce false results. The antibiotic-impregnated disks are carefully placed onto the surface of the bacterial lawn of growth and incubated. The agents will diffuse into the agar, establishing a concentration gradient, and if the organism is susceptible to the agent, a zone of clearing will appear around the disk where the growth has been inhibited by the antibiotic. The size of the zone of inhibition is measured and it depends on the bacteria to the specific antimicrobial agent and the point at which the antibiotic's minimum inhibitory concentration (MIC) is reached. The microbiologist must refer to the handbook showing the standard ranges and the low, medium and high MIC's and record these readings based on the standards for reliability.
The agar used is Mueller-Hinton based with a pH of 7.2-7.4 poured to a depth of 4 mm in 100 or 150 mm Petri dishes. The depth has an effect on the diffusion rate, so plates must not be expired and must be checked to make sure the depth is correct. If the agar is too thick, the diffusion rate will slow down and produce smaller zones. If the agar is too thin, the diffusion rate will speed up and produce larger zones.
The disks contain a specific amount of antibiotic and will be printed on the disk itself. Disks can be put into a disk dispenser to uniformly deposit them across the surface of the agar. Sterile tweezers may also be used to carefully place the disks on the surface, being careful not to touch the agar or bacterial lawn, then tamp them with the tweezers to make sure they stick and stay in place. It should be incubated at 35 degrees Celsius for 16-18 hours. The zones around each antimicrobial disk are typically uniform and even, however, a synergistic effect of two antibiotics may occur, producing a clear area between the disks, meaning that they are bactericidal in combination with each other.
The agar used is Mueller-Hinton based with a pH of 7.2-7.4 poured to a depth of 4 mm in 100 or 150 mm Petri dishes. The depth has an effect on the diffusion rate, so plates must not be expired and must be checked to make sure the depth is correct. If the agar is too thick, the diffusion rate will slow down and produce smaller zones. If the agar is too thin, the diffusion rate will speed up and produce larger zones.
The disks contain a specific amount of antibiotic and will be printed on the disk itself. Disks can be put into a disk dispenser to uniformly deposit them across the surface of the agar. Sterile tweezers may also be used to carefully place the disks on the surface, being careful not to touch the agar or bacterial lawn, then tamp them with the tweezers to make sure they stick and stay in place. It should be incubated at 35 degrees Celsius for 16-18 hours. The zones around each antimicrobial disk are typically uniform and even, however, a synergistic effect of two antibiotics may occur, producing a clear area between the disks, meaning that they are bactericidal in combination with each other.
e-test:
The E-test is a system to determine antibiotic sensitivity and determine the actual MIC. It is a paper strip with a gradient of antibiotic concentrations on one surface and the scale on the other surface. After preparing the KB plate with a McFarland standard solution for bacterial lawn of growth, the strip is placed antibiotic strip down on the surface of the agar with sterile tweezers. The plate is incubated and the antibiotic will diffuse into the agar to create an elliptical zone of inhibition. The point at which the inhibition zone intersects the scale on the strip is the MIC. If there is no zone of inhibition, this means the bacteria is resistant to the antibiotic.