bacterial capsules, slime layers, formation of biofilm, and mucoid morphotypes:
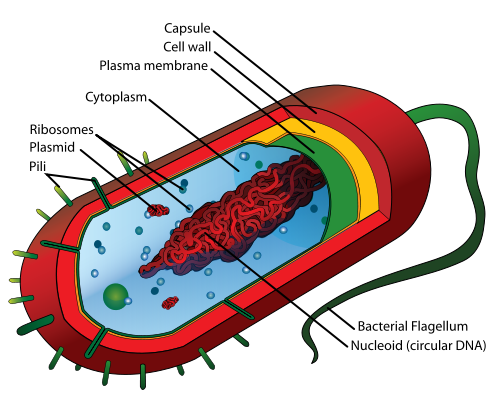
bacterial capsules:
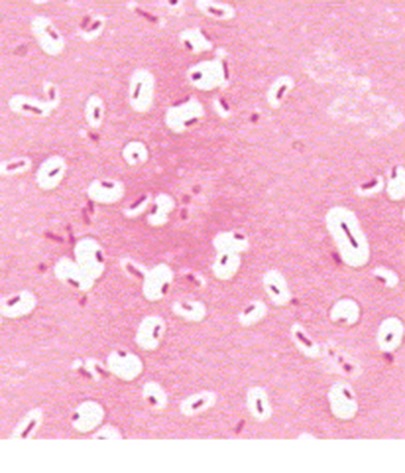
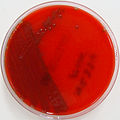
The bacterial capsule is a highly organized, thick layer of polysaccharide that is found surrounding the cell wall of some, not all, prokaryotes (bacteria). It is very adhesive and is not easily washed off by detergents or destroyed by centrifugation or antibiotics. The capsule can cause disease and infection, sometimes chronic infections, and is linked to relapses and to multi-drug resistance. Capsules are thick, the molecules are held tightly together, and they do not retain stain. Bacteria such as Klebsiella spp may have a clearing or halo around them as seen in the image below, indicating that a capsule is present. A special capsule stain may also be used for this purpose.
The bacterial capsule is a virulence factor since it has a variety of purposes for the cell, such as aiding in its protection from destruction and evasion of the immune system, contains and holds water to prevent the bacterium from drying out, thus promoting its survival, resist destruction by detergents, aid in adhesion to surfaces and to other cells for biofilm formation, and provide resistance to antibiotics.
A capsule is found in some, not all, bacteria, and includes some Gram-negative and Gram-positive bacteria:
The bacterial capsule is a virulence factor since it has a variety of purposes for the cell, such as aiding in its protection from destruction and evasion of the immune system, contains and holds water to prevent the bacterium from drying out, thus promoting its survival, resist destruction by detergents, aid in adhesion to surfaces and to other cells for biofilm formation, and provide resistance to antibiotics.
A capsule is found in some, not all, bacteria, and includes some Gram-negative and Gram-positive bacteria:
- E. coli
- K. pneumoniae
- P. aeruginosa
- N. meningitidis
- H. influenzae
- Salmonella spp
- S. aureus and MRSA
- S. epidermidis
- S. pyogenes
- S. agalactiae
- S. pneumoniae
- Some Bacillus spp
- B. fragilis
- S. maltophilia
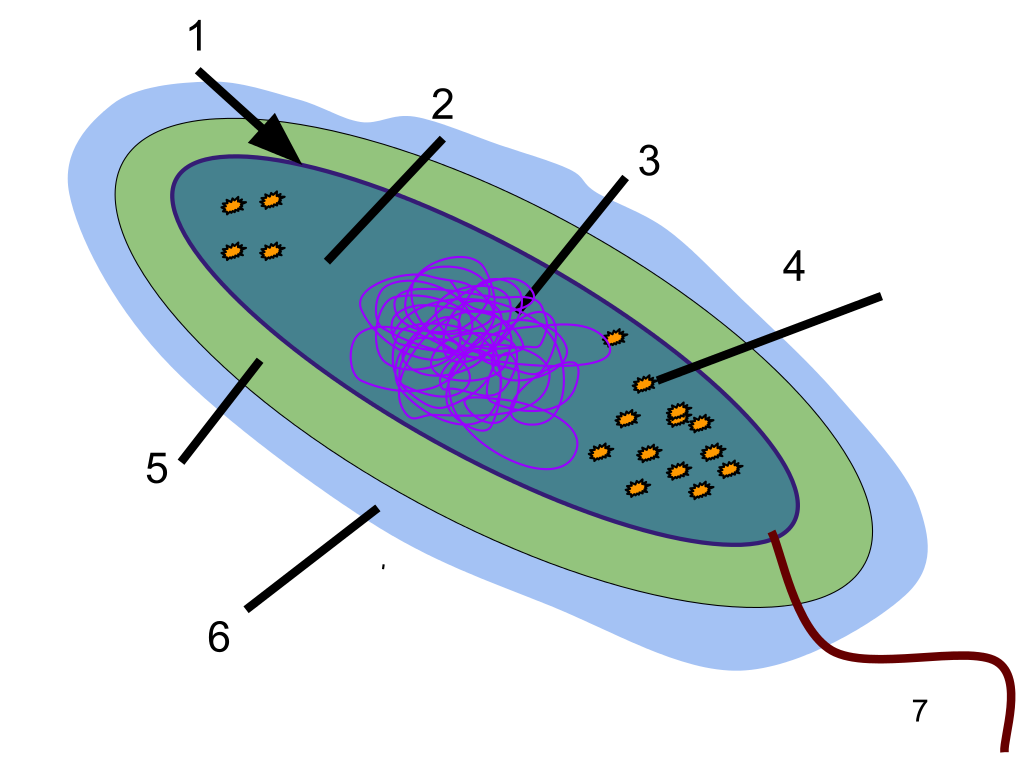
bacterial slime layer: glycocalyx
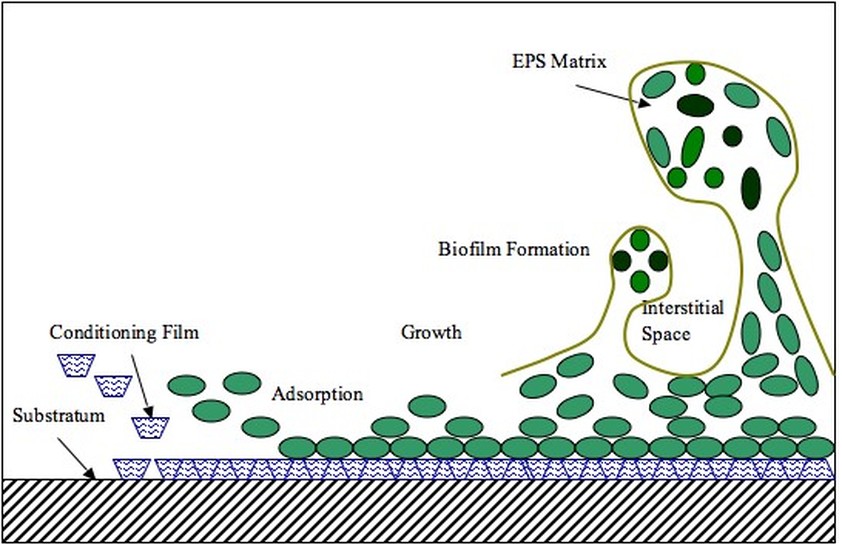
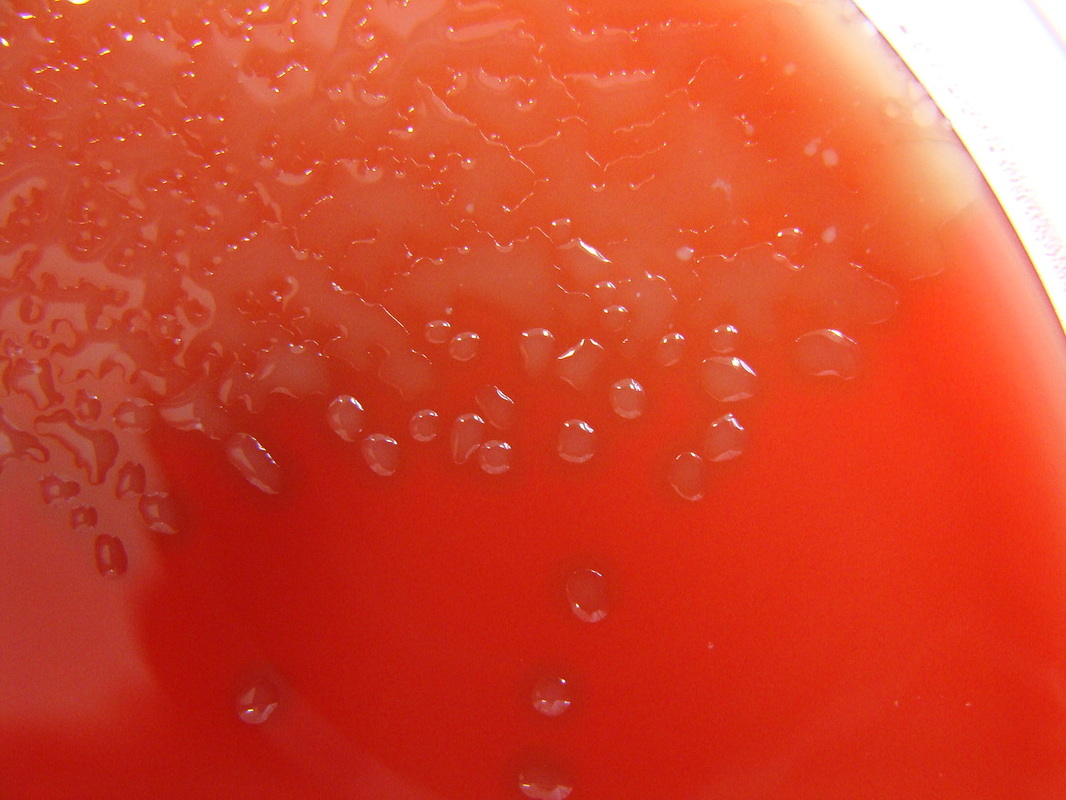
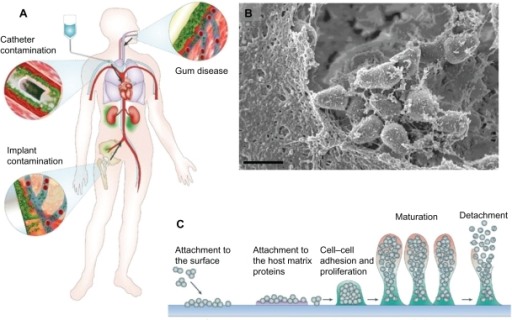
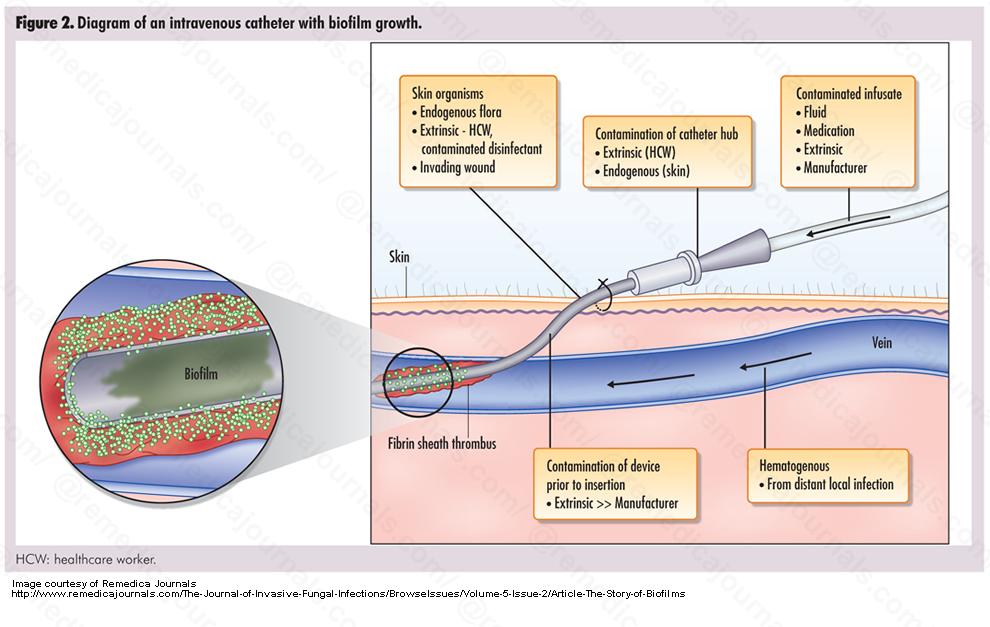
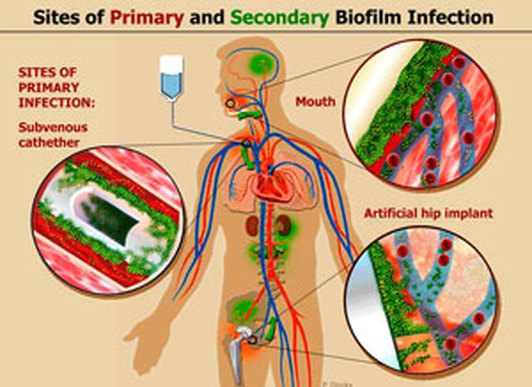
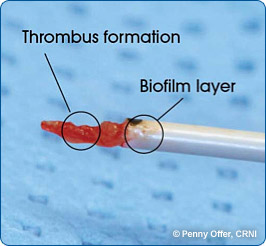
The slime capsule is made of glycocalyx and it is a viscous, sticky, mucoid secretion of alginate that diffuses into the surrounding environment or medium. It is easily removed or destroyed by centrifugation or detergents, unlike the capsule. This unorganized layer of extracellular matrix surrounds the capsule of bacterial cells. It consists of glycoproteins, glycolipids and exopolysaccharides (alginate). It is moist and protects the bacteria from drying out. It also protects it from destruction by the immune system or by antibiotics. It is the cause of the formation of bacterial biofilms in CF patients and on surfaces such as IV or urinary catheters or prosthetic devices or artificial heart valves. It enables bacteria to become "extremophiles", surviving harsh chemicals, extreme temperatures, changes in pH, changes in osmolality, and other changes or extremes of environment.
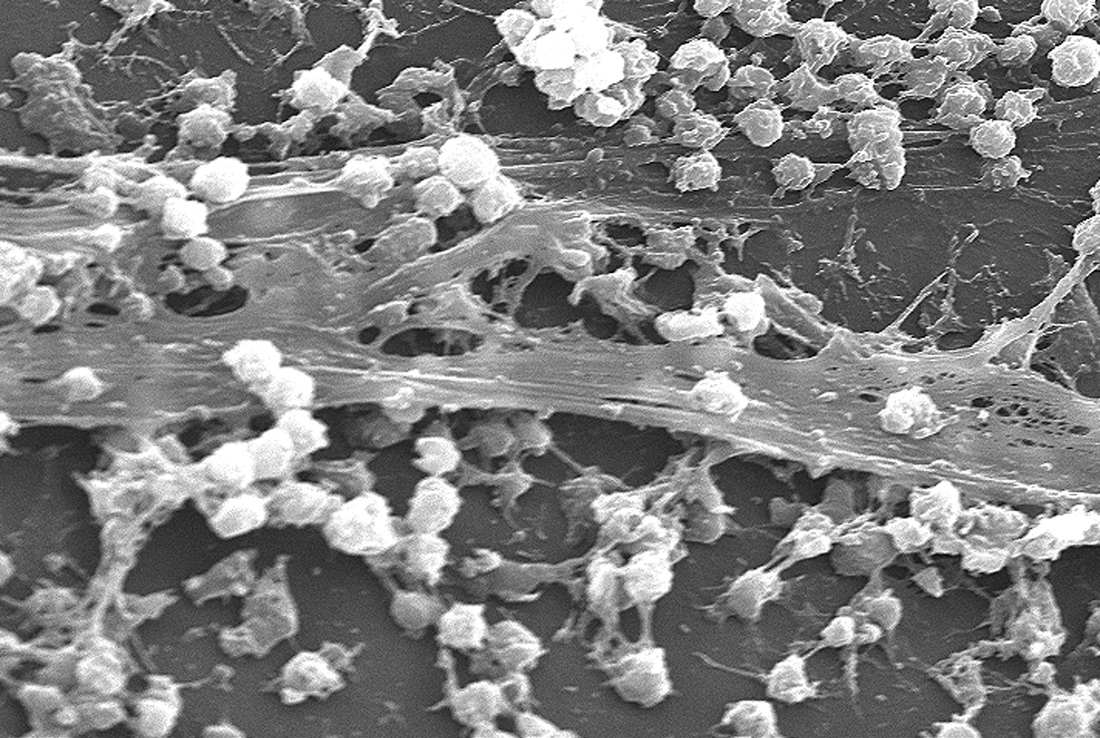
bacterial biofilms:
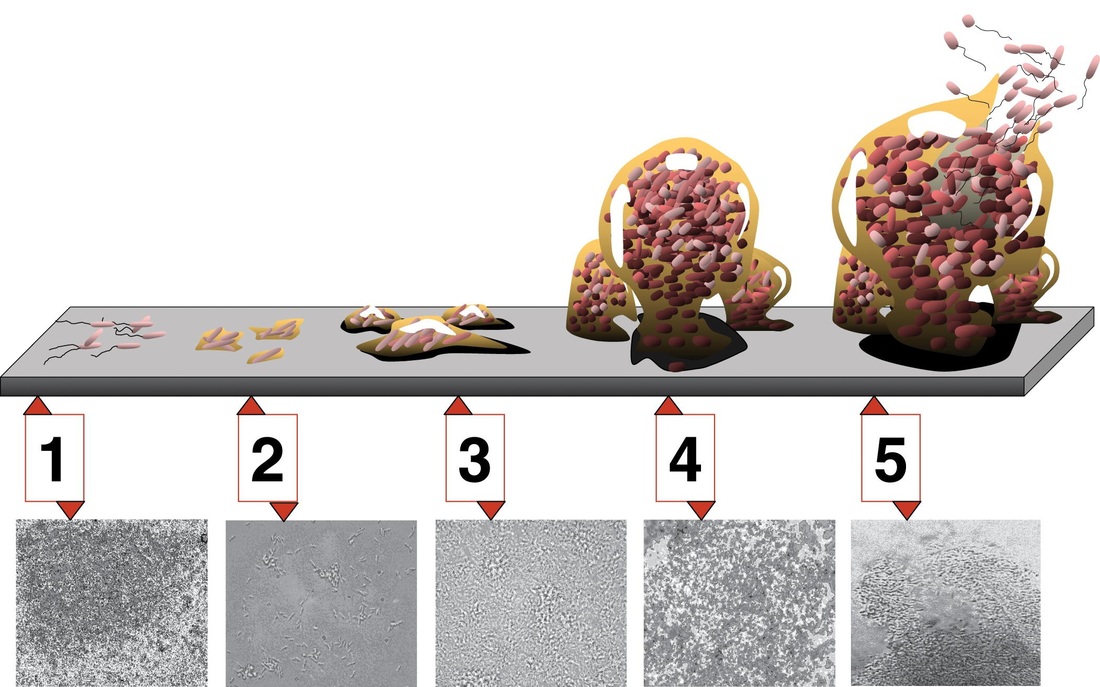
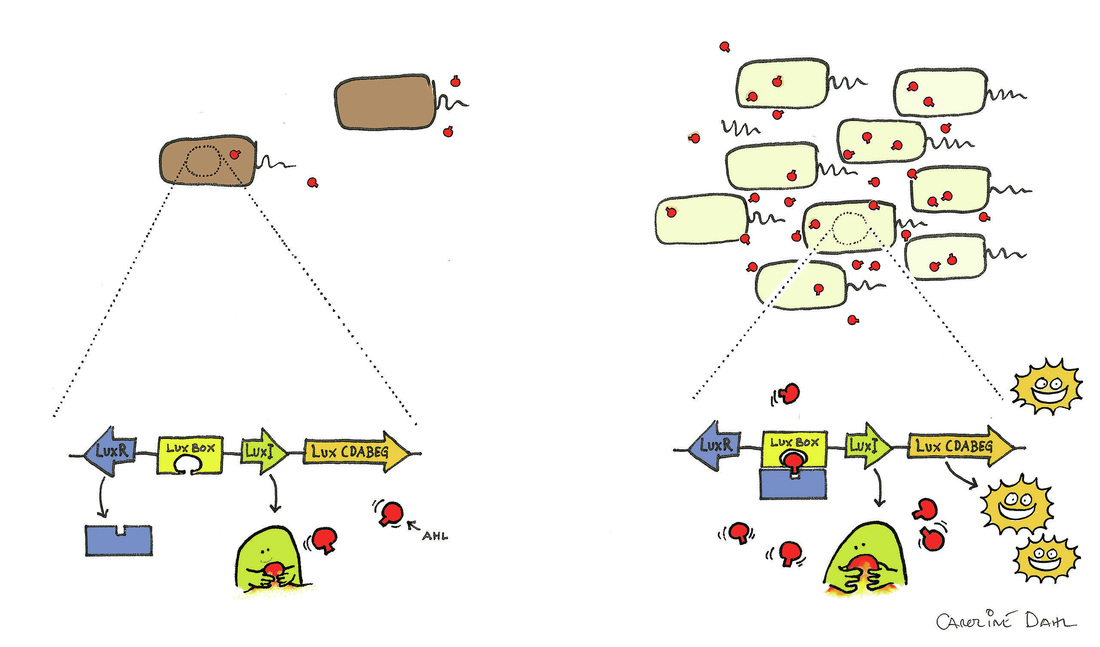
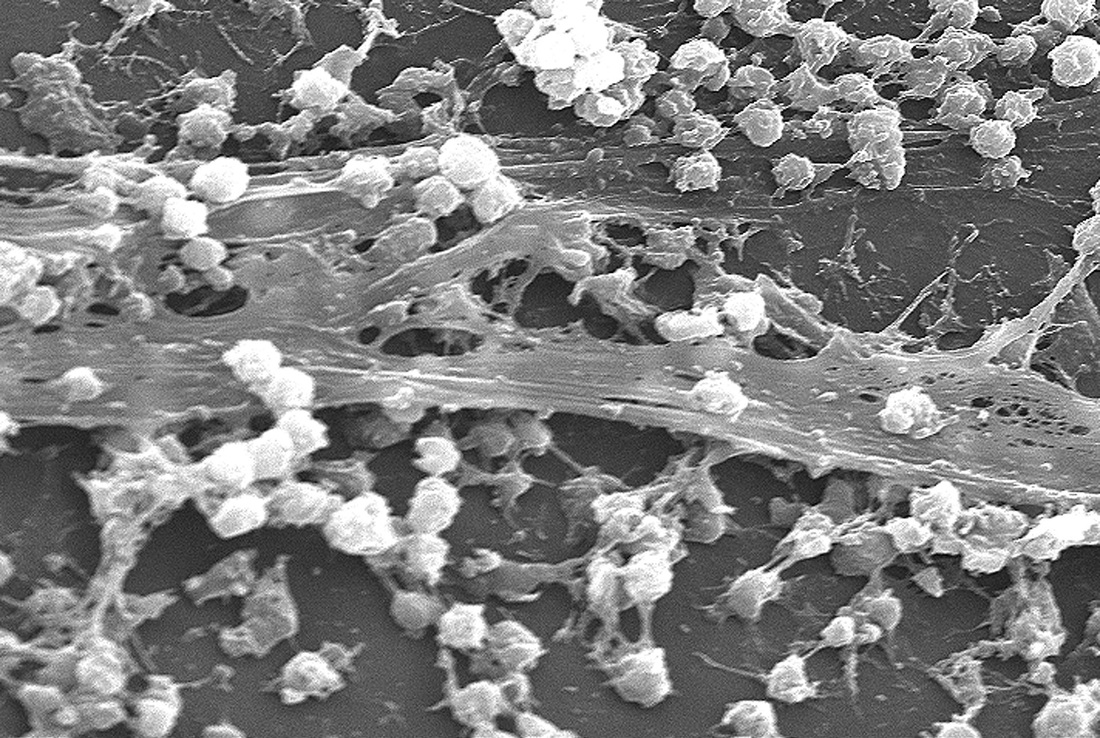
A biofilm is a matrix that is formed as microbes stick to each other and stick to surfaces via expression of adhesive molecules. This matrix is called "EPS", or extracellular polymeric substance. It is also called slime, which is why bacteria with slime layers are prone to form these biofilms for survival. This consists of polysaccharides, DNA and proteins. Cells literally "switch" or "shift" to a biofilm mode of growth via mass gene expression by genes that are highly and specifically regulated. This occurs by quorum sensing, a system of signaling (bacterial communication and response) linked to population density (molecular weight or light). This is used to coordinate gene expression. P. aeruginosa, for example, uses this method to coordinate biofilm formation, swarming motility, virulance factors, cell adhesion, and the production of polysaccharide. Once a threshhold is reached, the bacteria increase in virulence and become aggressive, in order to cause infection, evade the immune system, and protect themselves from antibacterial destruction. The genes Luxl and LuxR are responsible for this formation.
mucoid morphotypes and examples:
Pseudomonas aeruginosa begins as a non-mucoid phenotype/morphotype in Cystic Fibrosis patients, however, once colonization takes place and the bacterium becomes permanently established, only then does it nearly always mutate into a mucoid strain. Why does this occur? Well, it is similar to "survival of the fittest". The bacteria come together and signal each other through "quorum sensing" to form a bacterial biofilm that evades the immune system, entraps enzymes, promotes
"persisters", or a few dormant cells that survive antimicrobial therapy, detergents, chemicals, and extremes of environment and changes by enabling some non-growing or slow-growing individual microbes to serve as a reservoir for chronic and relapsing infections. This is a very complex process that involves a genetic component, as well as processes utilized by the bacteria to evade destruction and to "vomit out" antibiotic therapy via efflux pumps.
Because CF patients have abnormally increased levels of Na+ (sodium) and Cl- (chloride) and Ca2+ (calcium) in their respiratory tract, their respiratory fluids/secretions are very thick, viscous, sticky, adhesive, and create a high osmolarity in the lungs. A very mucoid substance called alginate is secreted, promoting an environment welcoming to bacterial colonization and chronic infection due to these conditions. Cystic Fibrosis is an autosomal-recessive genetic disorder characterized by disturbances in the electrolyte transport system and balance and disturbances in mucus secretion from the exocrine glands and secretory cells with atypical glycoproteins that are hyperviscous or thick. This leads to obstruction of ducts throughout the body, especially the respiratory tract. Therefore, there is an abnormal accumulation of abnormal fluids in the respiratory tract of CF patients. This leads to a decrease in the diffusion of oxygen, making it difficult to breathe.
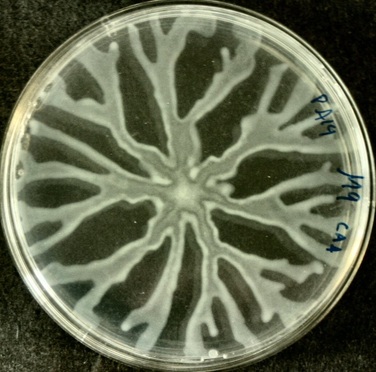
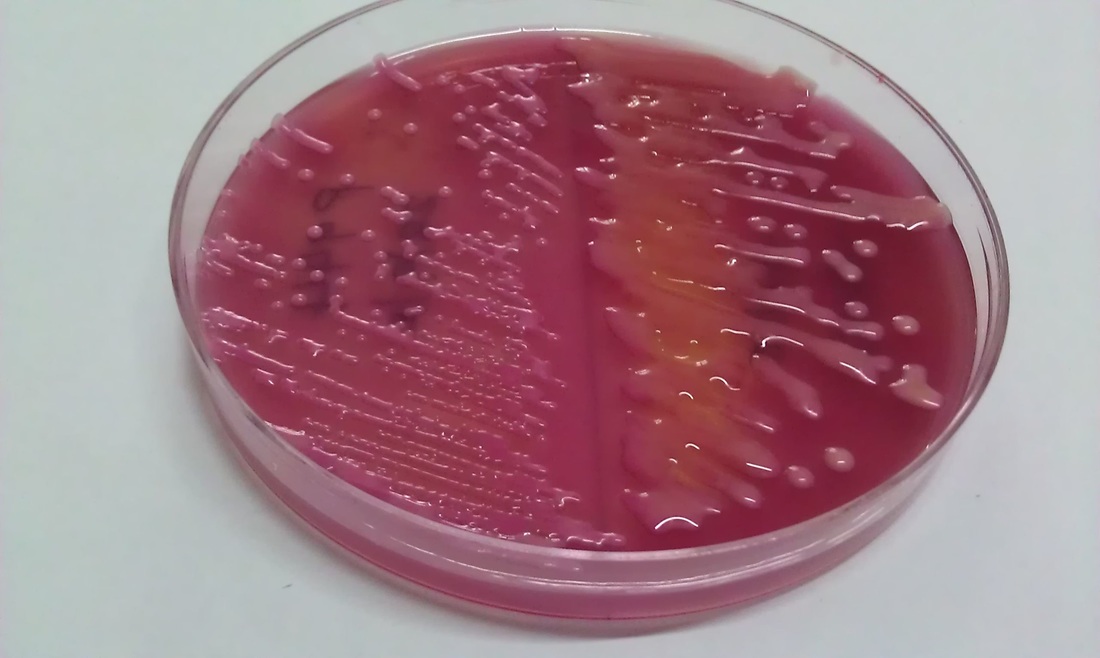
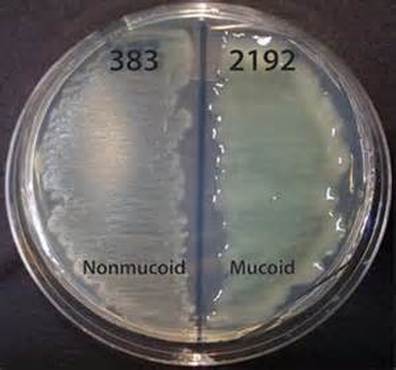
Initially, strains of the colonizations of P. aeruginosa start out as nonmucoid, but prolonged colonization or infection leads to a shift to the mucoid strain, which occurs genetically in late-stage CF. Large amounts of the slimy exopolysaccharide alginate lead to an extra thick mucus which promotes and supports bacterial biofilm formation within the lungs. This occurs in chronic/long-term/ongoing infections and colonization. The mucoid strain has adapted to a new environment and is often multi-drug resistant (MDRO). This occurs by expression of the pel and psl genes, which code for the adhesins pilin and flagellin, aiding in the stickiness and swarming motility of this bacterium. Another virulence factor is the formation of a slime layer secreted from the bacterial capsule, made of glycocalyx. The bacteria signal each other and communicate via signaling molecules that detect changes in the light or molecular weight of the environment. They signal each other to "join" the crowd and stick together to form a matrix called a biofilm. This matrix is an EPS matrix made up of nucleic acids, amino acids, carbohydrates, and ions. This forms a sort of "mesh" that entraps enzymes needed for both biosynthesis of more alginate, as well as enzymes that break the matrix down to release individual microbes so that they can spread and go form colonizations elsewhere in the body. These are the "persisters". The new genetic information is shared with new bacterial generations via binary fission, horizontal transfer (conjugation), mutations, and integrons. The genome of P. aeruginosa is the largest resistance, with >50 resistance genes to antimicrobials! Many strains are ESBL-producers (mucoid morphotypes), and they mutate repeatedly.
The mucoid phenotypes/morphotypes contain slime layers that are rich in glycoproteins, glycolipids and exopolysaccharides (alginate). This leads to the thick, viscous, shiny, slimy, stringy, wet, sticky, adhesive colonies you see on the plate of a mucoid strain. The mucoid phenotypes cloak antigens on the surface of the bacterial cell that evade the immune system leading to antiphagocytosis, strong attachment to the mucosal epithelium of the upper respiratory tract, increased morbidity (illness) and mortality (death), increased severity of pulmonary infection, and multi-drug resistance, since the bacterial capsule and slime layer is extremely difficult to penetrate, therefore, patients with these strains are very difficult to treat and are prone to relapses and chronic infections. The slime layer inhibits macrophage phagocytosis, so the bacteria survives extremes. It evades complement, so the long-term antibody memory may fail. This is why these infections are so difficult to treat.
"persisters", or a few dormant cells that survive antimicrobial therapy, detergents, chemicals, and extremes of environment and changes by enabling some non-growing or slow-growing individual microbes to serve as a reservoir for chronic and relapsing infections. This is a very complex process that involves a genetic component, as well as processes utilized by the bacteria to evade destruction and to "vomit out" antibiotic therapy via efflux pumps.
Because CF patients have abnormally increased levels of Na+ (sodium) and Cl- (chloride) and Ca2+ (calcium) in their respiratory tract, their respiratory fluids/secretions are very thick, viscous, sticky, adhesive, and create a high osmolarity in the lungs. A very mucoid substance called alginate is secreted, promoting an environment welcoming to bacterial colonization and chronic infection due to these conditions. Cystic Fibrosis is an autosomal-recessive genetic disorder characterized by disturbances in the electrolyte transport system and balance and disturbances in mucus secretion from the exocrine glands and secretory cells with atypical glycoproteins that are hyperviscous or thick. This leads to obstruction of ducts throughout the body, especially the respiratory tract. Therefore, there is an abnormal accumulation of abnormal fluids in the respiratory tract of CF patients. This leads to a decrease in the diffusion of oxygen, making it difficult to breathe.
Initially, strains of the colonizations of P. aeruginosa start out as nonmucoid, but prolonged colonization or infection leads to a shift to the mucoid strain, which occurs genetically in late-stage CF. Large amounts of the slimy exopolysaccharide alginate lead to an extra thick mucus which promotes and supports bacterial biofilm formation within the lungs. This occurs in chronic/long-term/ongoing infections and colonization. The mucoid strain has adapted to a new environment and is often multi-drug resistant (MDRO). This occurs by expression of the pel and psl genes, which code for the adhesins pilin and flagellin, aiding in the stickiness and swarming motility of this bacterium. Another virulence factor is the formation of a slime layer secreted from the bacterial capsule, made of glycocalyx. The bacteria signal each other and communicate via signaling molecules that detect changes in the light or molecular weight of the environment. They signal each other to "join" the crowd and stick together to form a matrix called a biofilm. This matrix is an EPS matrix made up of nucleic acids, amino acids, carbohydrates, and ions. This forms a sort of "mesh" that entraps enzymes needed for both biosynthesis of more alginate, as well as enzymes that break the matrix down to release individual microbes so that they can spread and go form colonizations elsewhere in the body. These are the "persisters". The new genetic information is shared with new bacterial generations via binary fission, horizontal transfer (conjugation), mutations, and integrons. The genome of P. aeruginosa is the largest resistance, with >50 resistance genes to antimicrobials! Many strains are ESBL-producers (mucoid morphotypes), and they mutate repeatedly.
The mucoid phenotypes/morphotypes contain slime layers that are rich in glycoproteins, glycolipids and exopolysaccharides (alginate). This leads to the thick, viscous, shiny, slimy, stringy, wet, sticky, adhesive colonies you see on the plate of a mucoid strain. The mucoid phenotypes cloak antigens on the surface of the bacterial cell that evade the immune system leading to antiphagocytosis, strong attachment to the mucosal epithelium of the upper respiratory tract, increased morbidity (illness) and mortality (death), increased severity of pulmonary infection, and multi-drug resistance, since the bacterial capsule and slime layer is extremely difficult to penetrate, therefore, patients with these strains are very difficult to treat and are prone to relapses and chronic infections. The slime layer inhibits macrophage phagocytosis, so the bacteria survives extremes. It evades complement, so the long-term antibody memory may fail. This is why these infections are so difficult to treat.
pseudomonas aeruginosa: mucoid morphotype seen in cystic fibrosis patients
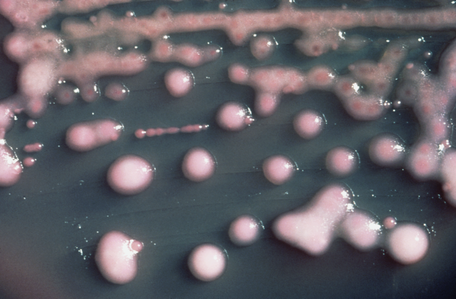
streptococcus pneumoniae: mucoid morphotype
other examples of bacterial biofilm producers:
- Staphylococcus epidermidis
- Staphylococcus aureus and MRSA
- Enterobacter spp
- Citrobacter spp
- Bacillus spp
- Listeria monocytogenes
- Lactobacillus
- Legionella spp
- E. faecalis
- Proteus spp
- Morganella morganii
- Providencia spp
- Other Coagulase Negative Staph